All published articles of this journal are available on ScienceDirect.
Correlation of Some Trace Elements Serum Levels with Prostate Cancer Progression in Saudi Patients
Abstract
Background:
Trace elements, such as zinc, arsenic, cadmium and nickel are found naturally in the environment, and human exposure comes from a variety of sources, including air, drinking water, and food. Yet, there are a few studies of the association between trace element levels and prostate cancer in the country.
Objective:
This study aimed to investigate the changes in trace elements in prostate cancer patients with different levels of their prostate-specific antigen (PSA) values.
Methods:
The study included 58 patients with prostate cancer aged 70 years and older, divided into 3 different levels of PSA. Full history and clinical data were recorded for all subjects. Blood samples from all subjects and levels of Se, Zn, Cd and Cu were analyzed by inductively-coupled plasma mass spectrometry. The odds ratio of trace element levels was adjusted in accordance with socioeconomic data, family history and supplements intake.
Results:
Mean Se and Zn levels in serum were significantly low (p<0.05) in all prostate cancer patients. The levels of serum Se decreased by 56%, 67% and 70%, while the levels of serum Zn decreased by 35%, 41% and 47%, in subjects with PSA of 5-10 ng/ml, 11-20 ng/ml and > 20 ng/ml, respectively. Cu levels were increased significantly in prostate cancer patients, while Cd levels had no significant difference between control and prostate cancer groups.
Conclusion:
This study emphasizes the importance of minerals intake during prostate cancer management and follow-up period. This highlights the importance of trace elements Zn and Se intake as food supplements for prostate cancer patients.
1. INTRODUCTION
Recent studies defined environmental factors as the risks of prostate cancer [1]. Environmental exposure has had implications on prostate cancer evolution or prevention [2]. Trace elements, such as zinc, arsenic, cadmium and nickel are obviously found in the surrounding environment, and human exposure comes from different sources, including air, drinking water, and food [3]. World Health Organization has categorized 19 trace elements as essential to human health, including arsenic (As), cadmium (Cd), nickel (Ni), selenium (Se), and zinc (Zn) [4]. Role of heavy metals in cancer progression is still unexplained [5]. Arsenic levels have been found linked with cancer risk, focusing on the detection from drinking water. Several articles discussed Cd and Ni exposure levels in different workplaces. Also, the role of Zn in several cancer sites has been examined as well, while Se did not show a direct relation to cancer risk [6] Trace elements are essential biological structures. Sometimes, these can be toxic at levels beyond those required for their biological functions. The toxicity increased with other nonessential elements of similar atomic structures that can imitate their activity of a trace element [7].
Trace elements are existent in the human body in very small levels, but they act dynamically in many biochemical enzymatic interactions and have been evaluated to have a potential in different diseases involving cancer [8]. Some trace elements may have a role in the growth of cancer cells by producing free radicals against oxidative stress which contributes to cancer growth [9]. Many studies showed the important effects of trace elements as segments of many enzymes that highly affect many biological processes. They have some regulatory functions that may affect immune reactions and free radical generation [10, 11]. The cancerous tissue directly indicates trace elements variations that are affected by the disease [12]. Trace elements concentration in the human body differs on the basis of the diet, sex, age, individual demand for each trace element, socioeconomic conditions, the content of chemical elements in drinking water, geographical location, and environmental pollution [13]. Many studies showed that levels of some minerals are highly associated with pathological disorders [14]. Recent studies showed the importance of trace elements cycle in blood and its role in the initiation of cancer, confirming their use as biomarkers [15]. Nowadays, trace elements are used as tumor biomarkers in different types of cancers [16]. Other authors showed also that alterations of elements in plasma and cancer tissues were observed in patients with, colorectal cancer [17, 18], breast tissues [19], malignant prostate [20], ovarian tissues [21], head, and throat cancers [22]. Many epidemiologic and biochemical studies have shown a potential correlation between human mineral levels and prostate cancer incidence. A list of these minerals included B, Ca, Mg, Se, Co, Cr, Fe, Mn, Zn [23, 24]. Unfortunately, the results of the intake of minerals levels on prostate cancer risk are inconsistent [25]. Some studies proved a positive association, while other articles report inverse proportion or even no association [26].
However, there is evidence of mineral bioavailability decrease in the elderly [27]. The theory of deficiency of minerals in the human body explained this phenomenon to be a result of lifestyle changes, dietary habits, and physiological effects of aging [28]. The elderly male has different deficiency levels of Cu, Fe, Mn, Se, Zn and antioxidants that increase the prostate cancer risk [29]. On the other hand, other epidemiologic studies have looked at potential connections between risk and mineral overloads resulting from excessive use of dairy foods (major source of Ca), red meat (a major source of Fe and Zn), and mineral supplements (Ca, Mg, Fe, Zn, Se and others) [30, 31].
1.1. Prostate Cancer and Body Trace Elements
Prostate Cancer and Cadmium level in the human body are introduced through food contamination (soil fertilizers), intake of grains, oysters and occupational exposure [31-33]. Recent studies showed a positive association with elevated levels of prostate cancer [34]. While selenium and Zinc were shown as protective factors of prostate cancer. Diet such as meat, egg and seafood has high concentrations of selenium [35]. Studies showed an inverse association between Se and prostate cancer mortality [36]. Many studies found that men taking Se as supplements rather than food for a long period had a significant reduction of the prostate cancer incidence of about 65% [37, 38].
Zn is an essential element present in meat, grains, dairy and vegetables. It is a major component in metalloenzymes which is essential for cell development and recurrence, osteogenesis and immunity. Zn is a major antioxidant in different cell types. Some studies have proven an association of lower Zn intake in prostate cancer patients', while other studies demonstrated no association [39]. Zn is known as a cellular growth defender in prostate cancer. Zn high levels are accumulated in human prostate [40]. In contrast, Zn concentrations in prostate cancer cells are significantly decreased than levels found in non-prostate tissues [41]. Studies demonstrate that Zn suppresses cancer growth in prostate progression; most likely due to the stimulation of cell cycle detention and apoptosis [42]. It was explained that the loss of high levels of Zn is a leading factor in the progression of malignant prostate cells [43]. In-vitro Zn maintains an intra-prostatic balance of testosterone and dihydrotestosterone - DHT [44].
1.2. Global Incidence of Prostate Cancer
Prostate cancer is the second predominantly mutual triggered in male cancer-related deaths and the most prevalent male non-cutaneous malignancy in the Western world [45]. PCa is the fourth predominant type of cancer worldwide [46]. Prostate cancer is known as the second common types of cancer in males worldwide with approximately 1,111,700 annual new cases [47] and the sixth common male cancer in Saudi Arabia.
In Saudi Arabia, King Faisal International Agency for Cancer Research (IARC) estimated that the age-standardized incidence ratio (ASIR) for prostate cancer accounted for 7.7 per 100,000 men in 2008, and the age-standard mortality rate (ASMR) was 5.1 of 100.000 men. Moreover, in 2011, King Faisal Research registry recorded 1231 cases (1.7%) of these cases are of prostate cancer admitted to the hospital during the years 1975-2011.
This study aimed to investigate the alterations of some trace elements in prostate cancer patients with different levels of their prostate-specific antigen (PSA) values [48].
2. MATERIALS AND METHODS
This study included 58 prostate cancer patients (mean age 71.1±5.4 years). Subjects were grouped in accordance to their PSA values into three: low grade PSA (n= 20) (5-10 ng/ml), medium grade (n=23) (11-20 ng/ml) and high-grade PSA (n=15) (>20 ng/ml). 30 healthy individuals had a mean age of 60.0±5.1 years. Healthy men were randomly selected among volunteers of matched socioeconomic status that did not show known disease and had no history of drug abuse. The available clinical data were reviewed for each subject. All the subjects were free of any intersex conditions, endocrine disorder or any other chronic diseases that may affect normal prostate development. All research subjects were confirmed for not receiving any mineral supplements that may affect prostate morphology. Patients yielded digital rectal screening, trans-rectal ultrasonography and pelvis CT scanning, bone scanning, and histopathological examination to assess metastatic disease and determine disease stage.
2.1. Samples Collection
A 10 mL blood sample was drawn in the morning after overnight fasting from healthy subjects and one week after digital rectal examination for patients with prostatic diseases. Serum of blood samples was separated and stored at -20 °C in a deep freezer until the date of analysis. Blood samples were drawn into containers with EDTA and another portion in plain centrifuge tubes and were allowed to clot,and then spun at 3000 rpm for 15 minutes. Serum levels of total (tPSA) were measured by chemiluminescence ELISA technique IMMULITE, DPC (Diagnostic Products Corporation, Los Angeles, CA, USA).
2.2. Laboratory Analysis Methods
Trace elements analysis (Cd, Zn, Cu and Se) concentrations were evaluated by ICP-MS (Perkin Elmer 7300, Perkin Elmer, USA), according to manufacturer's instructions. The operating conditions were as follows: 1) Carrier gas (argon, 99.999%), 0.8% L/mints; 2) Plasma gas (argon, 99.999%), 13 l/mints; 3) Auxiliary gas (argon, 99.999%), 0.8 l/min; pump rate, 1.5 ml/mints and power 1055 KW. The recovery yields of metal elements were higher than 95%. The detection limits of Hg were higher than 95% with detection value ≤3 ng/m3. The maximal value of relative standard deviation for the three replicates analyses of every individual sample was less than 4%.
Quality assurance and control (QA/QC) was carried out for all samples by analyzing with control sample regularly to ensure reliability; reproducibility and linearity were determined for each analysis. A linear calibration curve was obtained by using a blank and five-point calibration curve with different concentrations: 0.01, 0.2, 0.5, 1.0 ppm for all trace elements.
To check the instrumental errors, high purity ICP Element Standard Solutions VI CertiPUR 10 mg/L from Perkin Elmer, USA was used for external calibration. All the solutions were prepared and stored in polypropylene vessels, which was cleaned prior to use by soaking in 10% HNO3 and then rinsed several times with ultra-pure water, which is produced by Millipore Mill-Q System (resistivity of 18.2 Ώ cm). All the calibration solutions were prepared daily at appropriate mass fractions as the samples to be analysed and in the same acid matrix, as the sample and blank solutions. The use of the same matrix for all solution preparations ensured that no additional variability or bias was introduced into the analytical determination from the acid content of the matrix (Table 1).
2.3. Statistical Analysis
Data are expressed as the mean ± standard deviation (SD) for different studied parameters and possible associations between them. Data between groups were compared using one-way analysis of variance (ANOVA), and the differences between the means of two out of three studied groups, were analyzed using an independent-sample t-test. The significance level was established at the P value of < 0.05. The validity (sensitivity and specificity), accuracy of each parameter and ratios thereof were calculated by area under the curve (AUC) in receiver operating characteristics (ROC) curve analysis. IBM SPSS Statistics v20 (IBM, USA) was used for the statistical analyses.
3. RESULTS
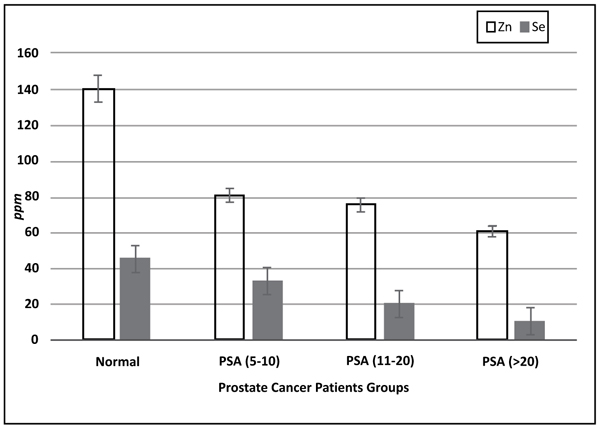
Table 1 showed demographic information and trace elements levels. No substantial difference was found between patients with prostate cancer and control subjects regarding age and BMI. Table 2 showed that Zn and Se concentrations were significantly less in prostate cancer patients and Cu levels were increased significantly in prostate cancer patients, while Cd levels had no significant difference between control and prostate cancer groups. Table 3 showed the variations of Cu/Zn ratio in prostate cancer patients. There was a considerable positive association between patients age and their PSA levels with different prostate cancer stages. PSA levels increased with the patient's age. Fig. (1) presents the Selenium and Zinc concentrations in prostate cancer subjects. Serum selenium and zinc concentrations were significantly below (p < 0.05) in all patients. Results showed that groups with PSA (5-10 ng/ml) had serum Se and Zn levels decreased by 56% and 35%. Whereas groups with PSA (11-20 ng/ml) concentrations of Se and Zn levels decreased by 67% and 41%. While Se and Zn concentrations decreased by 70% and 47% in groups with (PSA > 20 ng/ml)
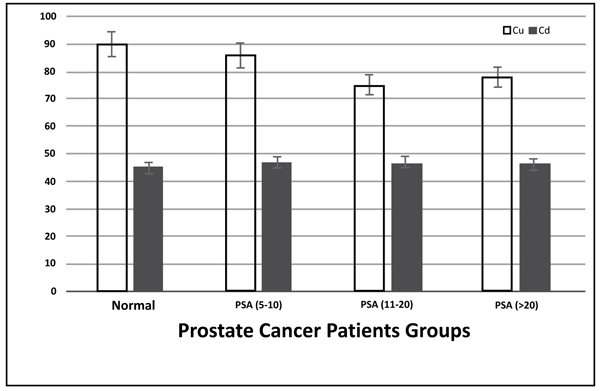
In Fig. (2), no significant difference was spotted (p>0.05) in serum concentrations of Cd of the subjects with prostate cancer with different PSA levels, as compared to control subjects.
| Instrumental Parameters | Data Acquisition | ||
|---|---|---|---|
| RF Power | 1400 W | Measuring mode | Segmented scan |
| Argon gas | flow 13 - 16 l/min | Point per peak | 5 |
| Nebulizer | 1.0 l/min | Scans/replicates | 3 |
| Plasma | 18.0 l/min | Replicate/sample | 3 |
| Sample | rate 190 s | Integration time | 398.6 s |
| Element | Control (n=30) | Patients (n=58) | P | ||
|---|---|---|---|---|---|
| Mean | St. Dev. | Mean | St. Dev. | ||
| Cd | 0.0011 | 0.001 | 0.0015 | 0.001 | 0.63 |
| Cu | 0.85 | 0.14 | 1.34 | 0.32 | 0.001 |
| Se | 1.93 | 0.32 | 0.53 | 0.25 | 0.001 |
| Zn | 2.64 | 0.56 | 0.73 | 0.41 | 0.001 |
| – | Subjects | |||
|---|---|---|---|---|
| Normal PSA <3 ng/ml | PSA (5-10) ng/ml | PSA (11-20) ng/ml | PSA (>20) ng/ml | |
| Mean age (years) | 60.0±5.1 | 70.1±2.4 | 74.1±2.4 | 75.1±4.4 |
| Cu/Zn Ratio | 0.67±0.05 | 0.83±0.06 | 1.21±0.12 | 1.34±0.83 |
4. DISCUSSION
Trace elements are major constituents of body's biological structures which have a complex role in the development and inhibition of cancer. Yet, the effects of high levels of trace elements on human health are questionable whether these elements have toxic effects when reaching higher concentrations than needed for biological functions or whether these elements are essential. Our study examined 58 prostate cancer patients aged 70 years and older with different PSA levels, and 30 normal subjects as controls aged 60s. Overall, serum selenium, zinc, cadmium and copper were analyzed in all subjects. There was a negative correlation between the trace element concentrations in serum and PSA levels assuring a high prostate cancer risk among all studied subjects.
Selenium has been known for its effective role in preventing cancers [49]. Different epidemiologic studies have approved the presence of negative association between selenium concentrations and prostate, lung and colorectum cancers [50]. These results were proven in our present study as well. However, after 4.5 years of treatment and 7.5 years of follow-up, further analysis showed a decrease in cancer progression risk. Prostate cancer patients who received selenium supplements were found to be with half selenium concentrations of the placebo group [51, 52]. Mechanisms include selenium role in arresting the cell cycle, decreasing cell proliferation, inducing apoptosis, DNA repair by p53 activation, disruption of androgen receptor indications, and acting as a core component of selenoenzymes [53], which integrates selenium as selenocysteine, an infrequently incident amino acid, to its active center [54]. The characteristics of selenocysteine are as unique redox with important antioxidant characteristics to selenoenzymes, as glutathione peroxidases and thioredoxin reductase, which are all expressed in the prostate [55]. Prostate cancer patients can be highly impacted by genes variations and superoxide dismutase (SOD) genes while receiving selenium supplementation.
The prostate is considered as a target organ for cadmium deposition. Some studies indicated that cadmium could be considered a carcinogenic factor in rats [56]. However, cadmium carcinogenesis mechanism is not completely clarified. Cellular and molecular mechanisms implied in cadmium carcinogenicity include proto-oncogene activation, tumor suppressor gene inactivation, disrupted cell adhesion and inhibition of DNA. In our study, Cd levels were found to be higher in patients with prostate cancer compared to controls, but the difference did not reach statistical significance.
Despite copper being an essential element for humans, high Cu concentrations may lead to cancer via toxic free radicals and producing DNA damage thus increasing serum Cu levels in several malignancies. Moreover, Cu levels could potentially cause progression of prostate cancer by improving blood supply to the tumor, which explains elevated Cu levels in involved tissues. Our results showed changes in serum copper levels among prostate cancer patients that were significantly different from normal (PSA < 3.0 ng/ml). The results agree with other studies [56].
It is known that Zn levels are significantly higher in prostate gland when compared to all tissues in the human body. Different studies indicated that Zn content increased in benign prostatic hyperplasia when compared to normal prostate tissue and that there was a decrease in prostate cancer, thus zinc levels are an important factor in the malignant prostate cells progression, which agrees with our study showing that serum zinc concentrations were significantly lower in prostate cancer patients than control subjects. Zn levels were the lowest in prostate cancer groups, while control groups were of the highest Zn levels. This observation agrees with the findings that zinc concentrations were lower in prostate cancer patients rather than Benign Prostate Hyperplasia (BPH) patients [57, 58]. Moreover, a significant difference was found in zinc concentrations in prostate cancer patients pre and post-therapy [59]. As Zinc plays a remarkable role in biochemical functions in the human body; it can simulate G2/M cell cycle phase arrest and apoptosis that is coupled with increased mRNA expression of p21 [60]. Zn facilitates early prostate cancer detection when the PSA value ranged from 4.1 ng/mL to 10.0 ng/mL. Serum Cu/Zn ratio could be an effective biomarker tool used for early detection rather than the free/total PSA ratio for prostate cancer early detection. However, the mechanism underlying Zn accumulation in prostate tissue and its importance are still unknown. Nevertheless, it was suggested that Zn deficiency could be a risk factor for prostate cancer [60]. In our study, it was demonstrated that there were significantly decreased Zn concentrations in patients with prostate cancer compared to the controls. Direct interference of Zn with intestinal absorption of Cu can result in higher levels of free Cu that can then displace Zn from metallothionein, because of copper’s high affinity for this protein. This postulated mechanism may explain the decrease observed in Zn concentrations that was accompanied by increased Cu concentrations measured in this study.
CONCLUSION
In conclusion, our study showed that there is an association between prostate cancer and trace elements. The study proves that increased levels of selenium and zinc can play important roles in the induction of prostate cancers. However, future prospective studies should address reasons for alterations in the serum concentrations of trace elements in patients with prostate cancer. Further prospective studies are needed to clarify the relationship between various stages of prostate cancer and serum levels of trace elements. Low levels of serum selenium and zinc in prostate cancer patients demonstrate the inter-relationship of these elements and their metalloenzymes. This study assures the significance of minerals intake during prostate cancer management and follow-up period. Moreover, this study may pose as a basis for recommending supplementation with selenium and zinc in the long-term management of prostate health.
AUTHOR'S CONTRIBUTIONS
All authors equally contributed to the article: the conception and design of the study, acquisition of data, analysis and interpretation of data, drafting the article and final approval of the submitted version.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study protocol was approved by the Ethics Review Board for Human Studies at the Faculty of Medicine, Umm Al-Qura University, KSA.
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was obtained from all patients and healthy subjects; the aim of the study and the procedures that would be required were described to them beforehand. All the subjects were assured that they could choose to discontinue their participation in the study without jeopardizing the medical care being given to them, including treatment and follow-up.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interests, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


