All published articles of this journal are available on ScienceDirect.
Diseases Coexisting with Sarcoidosis and Cost of their Treatment in Silesian Voivodeship (Poland)
Abstract
Objectives:
Sarcoidosis is a complex systemic disease with many symptoms of unknown etiology. The chronic course of sarcoidosis often allows it to co-occur with other diseases. A coexisting different form of sarcoidosis in some studies and no data regarding the costs of treatment justify the aim of the study. This study aims to assess a different form of sarcoidosis because no current data exist regarding the cost of this treatment.
Methods:
The epidemiological and economic analysis focused on diseases coexisting with sarcoidosis in the adult population of the Silesian Voivodeship (Poland), from 2011 to 2015, was conducted based on the secondary epidemiological data. The study involved 2,922 cases of the primary diagnosis of sarcoidosis (code D86 according to ICD-10) and 505 cases of sarcoidosis coexisting with another.
Results:
One out of ten patients with sarcoidosis has been found to be diagnosed with at least one comorbid disease. The comorbid disease includes diseases of the circulatory and respiratory system as well as endocrine, neoplasms, nutritional and metabolic diseases. The average unit cost of sarcoidosis therapy increases with the number of comorbidities and depends on the nature of the primary diagnosis and type of medical service.
Conclusion:
The epidemiological situation of diseases coexisting with sarcoidosis has been found to be similar to those reported in other European countries. However, neoplasms are one of the most common frequent comorbidities in Poland. The results obtained confirmed the strong impact of comorbidities on the cost of treatment of sarcoidosis.
1. INTRODUCTION
Sarcoidosis (code D86 according to International Statistical Classification of Diseases and Related Health Problems, 10th Revision – ICD-10) is a rare health problem classified as a multi-organ granulomatous disease. European studies on sarcoidosis epidemiology indicate a slight increase in its incidence and hospitalization rates. From 2006 to 2010, Poland observed an increase in the standardized incidence of lung sarcoidosis which ranged from 3.8/100,000 in 2006 up to 4.3/100,000 inhabitants in 2010 [1]. In a similar period (2002-2012), Switzerland observed a significant increase in crude hospitalization rate among the two age groups (45-58 and 70-96 years) within the range of 70-98/100,000 hospitalized (p<0.001), and 37-63/100,000 hospitalized patients (p<0.001), respectively [2]. Moreover, it was evident that when patients with sarcoidosis were rehabilitated, their hospitalizations were usually longer and the number of comorbidities was higher than other patients [2]. While the etiology of sarcoidosis remains unknown, its treatment is complicated due to the coexistence of other chronic diseases such as cardiovascular, autoimmune, chronic pulmonary, metabolic, chronic liver diseases, and neoplasms [3-5]. Swiss researchers showed that the risk of rehospitalization was significantly higher in patients with sarcoidosis with many coexisting diseases (HR=1.05; 95%CI:1.04-1.07) [2]. Polish observation confirmed that the risk of death in patients with sarcoidosis was significantly higher in the case of four or more comorbidities (HR=5.2; 95%CI:0.04-62.86) [5].
Moreover, the treatment of sarcoidosis is a serious economic burden for the state budget, and the number of comorbidities has a significant impact on the value of costs incurred [6, 7]. A USA study conducted in the years 1998-2015 showed that in the case of patients with the highest number of comorbidities along with sarcoidosis, the cost of services was the highest (80-100 percentile of total costs) [6].
No conclusive evidence regarding the impact of clinical forms of sarcoidosis and comorbidities on costs of treatment was our motivation for the study. The research aimed to assess the incidence of comorbidities among patients with sarcoidosis, including mutual assessment of its clinical form and the background of the epidemiological and economic situation.
2. MATERIALS AND METHODS
The secondary type of epidemiological data on sarcoidosis (D86, according to ICD-10) and data on coexisting diseases were obtained from the registry of the National Health Fund (NFZ) in Katowice. Data include the number of new cases of the disease, age, and sex of patients in the period 2011-2015. In the model of descriptive epidemiological research, we assessed incidence and occurrence of the following diseases: lung sarcoidosis (D86.0) and lung sarcoidosis with sarcoidosis of lymph nodes (D86.2), sarcoidosis of lymph nodes (D86.1), sarcoidosis of skin (D86.3), sarcoidosis of other and combined sites (D86.8) and finally sarcoidosis unspecified (D86.9). In the analysis, we used both the number of patients with primarily diagnosed sarcoidoses (an underlying diagnosis) and patients with coexisting sarcoidosis recognized as one of the threee most frequent comorbidities (a comorbid disease) in adult inhabitants (19+ years old) of the Silesian Voivodship. Moreover, analyzed data included both registered data at outpatient and stationary services.
In the calculation, we controlled the gender, the date of birth, the ICD codes of diseases registered as a primary diagnosis, or one of the three most frequent comorbidities, co-occurring with sarcoidosis. We presented the number and percentage of total sarcoidosis and also those clinical types of the disease for which the frequency of occurrence was at least 5%. Additionally, the assessment included calculations for the total population and separate age groups which are as follows: 19-34, 35-54, 55-64, and 65+ years. The same assessment was performed in the case in which sarcoidosis remained a coexisting disease with the recognized other underlying disease. The presented study was not an experiment; we used secondary, anonymous epidemiological data collected at the NHS registry. The study did not involve human or animal subjects, so the project did not need ethical approval.
The direct unit costs of sarcoidosis treatment, both outpatient and stationary treatment, were estimated in the years 2011 to 2015. We calculated the arithmetic mean of the treatment cost for patients with the first-time sarcoidosis recognized, including also the treatment of patients with comorbidities.
The statistical data analysis was performed using the procedures available in the MS Excel 2013 (Microsoft Office 2013). Quantitative variables are presented as means and standard deviation, and categorical variables are described by number and percentage.
3. RESULTS
The predominant types of sarcoidosis recognized as an underlying disease in adult (19+ years) inhabitants of the Silesian voivodeship in years 2011-2015 include lung sarcoidosis (n=1937; 66.3%), next sarcoidosis of lymph nodes (n=490;16.8%), unspecified sarcoidosis (n=323; 11%), while only 49 cases (1.7%) were patients with skin sarcoidosis. The average mean age of sick people was 46.6±13.3 years. The percentage of patients with at least one comorbid disease was 12.9%. Moreover, sarcoidosis was diagnosed in 505 patients as coexisting with another primary disease. Those patients were mostly hospitalized (Table 1).
| Sarcoidosis D86 as an Underlying Diagnosis |
Total number of new cases in 2011-2015 N (100%) |
Number and Percentage of Patients According to the Type of Treatment N (%) |
Number of Comorbidities N (%) |
Average age of Patients X±SD (years) |
|||
|---|---|---|---|---|---|---|---|
| O | H | 1 disease | 2 diseases | 3 diseases | |||
| Total | 2922 |
1609 (55.1) |
1313 (44.9) |
379 (12.9) | 153 (5.2) | 54 (1.8) | 46.6±13.3 |
| Sarcoidosis of lung D86.0, D86.2 |
1937 | 1133 (58.5) |
804 (41.5) |
227 (11.7) | 92 (4.7) | 31 (1.6) | 46.8±13.2 |
| Sarcoidosis of lymph nodes D86.1 |
490 | 215 (43.9) |
275 (56.1) |
73 (14.9) | 30 (6.1) | 12 (2.4) | 45.2±12.9 |
| Sarcoidosis of skin D86.3 |
49 | 29 (59.2) |
20 (40.8) |
10 (20.4) | 3 (6.1) | 2 (4.1) | 52±15.1 |
| Sarcoidosis of other and combined sites D86.8 |
123 | 61 (49.6) |
62 (50.4) |
29 (23.6) | 9 (7.3) | 3 (2.4) | 47.7±12.9 |
| Unspecified sarcoidosis D86.9 |
323 | 171 (52.9) |
152 (47.1) |
40 (12.4) | 19 (5.9) | 6 (1.9) | 46.8±13.6 |
| Sarcoidosis D86 as a comorbid disease |
Total number of new cases in 2011-2015 N (100%) |
N (%) | Comorbid diseases | Average age of patients X±SD (years) |
|||
| O | H | 1th | 2nd | 3rd | |||
| Total | 505 |
141 (27.9) |
364 (72.1) |
354 | 116 | 37 | 50.9±14.1 |
H – hospitalization, stationary treatment.
Among the most frequently coexisting diseases with sarcoidosis were cardiovascular (n=183; 31,2% of patients) and respiratory diseases (n=135; 23% of patients) (Supp. 1). For one in ten patients with sarcoidosis, endocrine, nutritional and metabolic diseases were diagnosed as comorbid diseases. Data indicate that the predominated diseases were hypertension, ischemic heart disease, chronic lower respiratory diseases, including COPD – 42 cases (7.2% of patients) and asthma – 40 cases (6.8% of patients), sarcoidosis in another form than the primary recognition, diabetes, disorders of the thyroid gland, arthropathies, urticaria, and erythema. Moreover, almost 5% of patients had symptoms of sarcoidosis (Supp. 2), including the following: cough, hemoptysis, abnormalities of breathing, pain in throat and chest, pleurisy (R04-R07 or R09) among 13 patients (2.2%), and enlarged lymph nodes (R59) among 10 patients (1.7%). Anxiety disorders (F41) were found to occur in only 2 people (0.3%).
Cardiovascular diseases were the most common diseases coexisting with sarcoidosis diagnosed in both women and men and the patients aged 65 and above (Supp. 3 and 4). In the case of female patients with lung or lymph nodes sarcoidosis, significant comorbidity was observed to frequently occur with endocrine, nutritional and metabolic diseases. However, in male patients, respiratory diseases were dominant. For example, significantly more thyroid diseases (17 out of 18 cases) were noted in women. However, the frequency of COPD was twice higher in men than in women. The pattern of comorbidities was similar in each age group of sarcoidosis patients, but cardiovascular diseases were most frequently diagnosed as a coexisting health problem in older patients (65+ years).
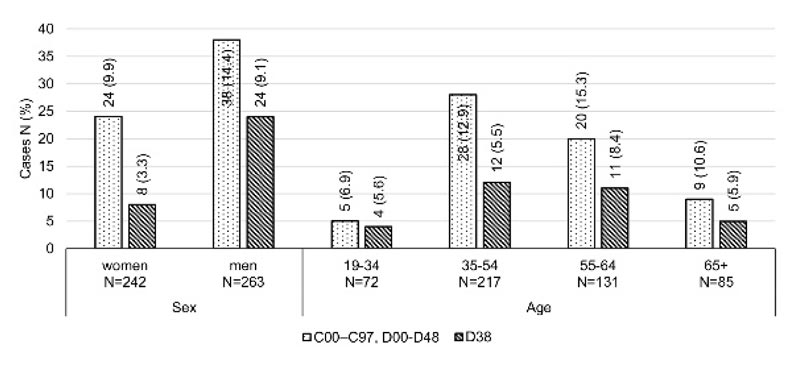
Due to the high frequency of coexisting neoplastic diseases and sarcoidosis, we decided to calculate the number and percentage of both diseases in separate demographic subgroups. The results of stratification suggest that the frequency of neoplasm of the middle ear and respiratory or intrathoracic organs was three times higher in males than females. The high frequency was also reported in the middle-aged group of patients, 35-64 years (Fig. 1).
The average unit direct cost of treatment of sarcoidosis patients granted by NFZ has been presented in Table 2. Hospitalization is more expensive than outpatient visits and direct costs rapidly increasing with the number of coexisting diseases.
| Type of Disease | The Average Direct Unit Cost of Treatment in the Study Period 2011-2015 [PLN / EUR] |
|||||
|---|---|---|---|---|---|---|
| Type of Treatment | Number of Comorbidities with Sarcoidosis | |||||
| Sarcoidosis D86 as the underlying diagnosis |
O | H | 0 | 1 | 2 | 3 |
| Total |
70.72 16.41 |
5 115.17 1 187.23 |
2 271.29 527.35 |
4 045.42 939.27 |
4 600.40 1 068.12 |
4 988.80 1 158.30 |
| Unspecified sarcoidosis | 69.94 16.23 |
4 665.93 1 082.96 |
2 460.68 571.32 |
3 039.05 705.61 |
3 661.88 850.22 |
4 525.50 1 050.73 |
| Sarcoidosis of other and combined sites |
52.03 12.08 |
4 996.67 1 159.73 |
2 265.91 526.10 |
4 188.68 972.53 |
4 698.08 1 090.80 |
8 060.00 1 871.37 |
| Sarcoidosis of lung | 72.89 16.92 |
5 235.45 1 215.15 |
2 117.67 491.68 |
4 307.53 1 000.12 |
4 793.10 1 112.86 |
4 938.36 1 146.59 |
| Sarcoidosis of skin | 36.73 8.53 |
1 846.95 428.68 |
715.12 166.04 |
1 015.93 235.88 |
1 716.00 398.42 |
1 716.00 398.42 |
| Sarcoidosis of lymph nodes | 69.83 16.21 |
5 276.25 1 224.62 |
2 912.93 676.32 |
4 159.71 965.80 |
4 807.15 1 116.12 |
5 640.29 1 309.56 |
| Sarcoidosis D86 as a comorbid diagnosis |
O | H | 0 | 1 | 2 | 3 |
| Total |
69.52 16.14 |
3 154.86 732.49 |
- |
2 137.99 496.40 |
2 515.90 584.14 |
3 267.64 758.68 |
H – Hospitalization, stationary treatment.
4. DISCUSSION
The epidemiological situation of sarcoidosis in Silesian Voivodeship varied in the study period. The most frequently registered form of the disease was lung sarcoidosis (66% of all diagnosed cases). An important observation is the coexistence of other diseases in patients with sarcoidosis. Among 12.9% of patients, sarcoidosis coexists with at least one more disease; the percentage is higher in patients with sarcoidosis of skin (20.4% of cases) and sarcoidosis of other and combined sites (23.6% of cases). The frequency of comorbidities reported by researchers from other countries is different and ranges from 29% to 90% [8-10]. It was confirmed that patients with multiorgan sarcoidosis had a significantly larger number of diagnosed comorbidities compared to patients with pulmonary sarcoidosis (1.52±1.47 vs. 0.9±1.1, p<0.05) [8]. On the other hand, data from Spain (N=218 patients with sarcoidosis in years 1990-2015) suggested that at least one coexisting disease was diagnosed in patients with sarcoidosis in comparison to the equivalent control group (50.5% vs. 29.4%; p<0.001) [4].
In this study, the most common comorbidities were cardiovascular diseases (31.2% of cases), respiratory diseases (23% of cases), and endocrine, nutritional and metabolic diseases (11.6% of cases), which is consistent with results from another part of Poland (N=1779) – 28.4%, 19.8%, and 12.2%, respectively [8]. Moreover, the dominant diseases were the following: hypertension (23.7% of patients with sarcoidosis), ischemic heart disease (4.9%), chronic lower respiratory diseases (14.2%), other diseases of the respiratory system (2,7%), diabetes (6.5%), disorders of the thyroid gland (3.1%) and obesity (1.5%). The results are similar to the incidence of the specified comorbidities sarcoidosis presented in other countries: hypertension (from 15% to 52.9% of cases with sarcoidosis), heart disease (2.3%-10%), asthma (2,2%-15%), COPD (2.3%-29.5%), bronchiectasis (1.1%-6.3%), ILD (29.9%), pneumonia (2.3%-15%), hyperlipidaemia (21.7%-41.5%), diabetes (5%-24.9%), thyroid disease (7%-13.1%), overweight (3.3%-14.2%) [4, 5, 8-12]. However, it should be noted that the observed comorbid diseases were coincidentally associated with the main health problems in the general Polish population [8]. On the other hand, Nowiński et al. [5] documented a significantly higher risk of thyroid disease in people with sarcoidosis in comparison to the control group (OR=3.62; p<0.05), while Brito-Zeron et al. [4] showed a significantly higher percentage of respiratory diseases among patients with sarcoidosis in comparison to the control group (9.6% vs. 2.3%; p<0.01). Also, results of Swiss study showed a significantly higher risk of hypertension (15% vs. 8.7%, OR=2.0, p<0.001), asthma (2.8% vs 0.8%, OR=3.7, p<0.001), pneumonia (2.3% vs. 0.6%, OR=4.1, p<0.001), COPD (2.3% vs 0.8%, OR=2.9, p<0.001), and diabetes (0.8% vs. 0.1%, OR=6.7, p<0.001) in patients with sarcoidosis compared to the control group [2]. The coexistence of sarcoidosis and asthma may be partially explained by the abnormal immune response related to environmental exposure [9].
A significant observation is the coexistence of distinguishing diseases of the musculoskeletal system and connective tissue with sarcoidosis (7.3% cases). Moreover, almost 6% of patients were diagnosed with other forms of sarcoidosis, while 3.2% of patients with urticaria and erythema, which mainly concerned those treated for pulmonary sarcoidosis and sarcoidosis of lymph nodes. Other studies also enumerated anemia (4.7%-19%), connective tissue disease (6.2%-8.2%), liver disease (10.6%) and vitamin D deficiency (1.1%-5.6%) [4, 5, 8-12].
Considerable attention is also given to the coexistence of sarcoidosis with cancers, the presence of which was registered in 0.9% to 10% of cases with sarcoidosis [4, 5, 8-12, 14]. Among mechanisms explaining such relationships are chronic inflammation, immunological disorders, and genetic susceptibility to both cancer and autoimmune diseases [14]. Uterus, testicles, liver, lung, skin, stomach, kidney are enumerated as the most susceptible organs. Moreover, the meta-analysis of 16 studies (N=27,268 patients with sarcoidosis, including 1,952 patients with cancer) revealed an increased risk of skin cancers (RR=2.0, 95% CI:1.7÷2.4), hematopoietic system (RR=1.9, 95%CI:1.4÷2.6), upper gastrointestinal tract (RR=1.7, 95%CI:1.1÷2.8), renal (RR=1.6, 95%CI:1.2÷2), liver (RR=1.8, 95%CI:1.03÷3.1) and large intestine (RR=1.3, 95%CI:1.1÷1.7) in patients with sarcoidosis [14]. There was no significant relationship observed between lung cancer and sarcoidosis (RR=1.2, 95%CI:0.8÷1.7) [14]. This study confirmed that neoplasms of the middle ear and respiratory and intrathoracic organs coexist with sarcoidosis in 7.1% of patients. The results obtained are in line with Agca's observation that cancer is related to middle-aged people below 50 years [9].
Earlier published data suggest that patients with lung sarcoidosis have typical symptoms such as cough, breathlessness, and fever. Those disorders frequently coexist with fatigue (23.6-90% of patients with sarcoidosis), depressive symptoms and anxiety (1.2-66%), sleep apnoea (2.4%-3%), arthralgia, muscle pain, and weakness [4, 5, 8-12, 15]. The occurrence and chronic nature of the disease have a significant impact on the quality of life, especially in daily activities, social and professional life [15]. The results suggest that symptoms of the disease were recorded among 4.6% of patients with sarcoidosis, including respiratory symptoms or enlarged lymph nodes among 3.9%, which confirms the nature of the disease in patients with pulmonary sarcoidosis (5.1%) and sarcoidosis of the lymph nodes (3.5%). Only 0.3% of the patients had anxiety, but no depressive behaviors were revealed. Meanwhile, current studies estimate the occurrence of anxiety disorders at the level of 33-36%, while only 6% of patients developed an anxiety attack [15, 16]. Moreover, the authors point to the importance of coexistence depressive and anxiety behaviors, both in the case of sarcoidosis and other chronic diseases, leading to negligence in pro-health activities (taking medicines, rehabilitation, and a healthy lifestyle) and decreasing quality of life [16, 17]. These symptoms are also associated with an increased sense of fatigue, which is more common in patients with multiple organ sarcoidosis and patients with additionally diagnosed hypertension or pulmonary hypertension, overweight, diabetes, and sleep apnoea [15, 18]. The relatively small percentage of mental and behavioral disorders recorded in Poland may be the result of applied diagnostic standards for interstitial lung diseases, including pulmonary sarcoidosis, in which specialists put emphasis on radiological and histopathological tests while omitting the assessment of emotional patients' condition and the quality of life [19].
The results confirmed the influence of sex and age on the incidence of diseases that coexisted with sarcoidosis [8, 9, 12]. Thyroid disease coexisting with sarcoidosis was most frequently in women than men; 95 (11.3%) vs. 29 (3.1%) and 81 (16.6%) vs. 10 (4.8%), respectively [8, 9]. In another study, a significantly higher risk of hypothyroidism in women with sarcoidosis than in control (OR=2.7, 95%CI:1.3-5.9) and significantly higher mean values of thyroid-stimulating hormone and AbTPO (antithyroid antibodies) were documented [20]. The coexistence of arthropathy reported in our study was twice frequently in the group of women, and such information is similar to Agca observation in which a significantly higher percentage of women with rheumatoid diseases, 20 (4.1%) vs. 1 (0.5%), was recognized [9]. The demographic structure of patients with sarcoidosis and COPD was similar to that reported in earlier studies [21]. The highest frequency of comorbidities for elderly patients (65+ years) was observed, which is in line with data reported by the Central Statistical Office among inhabitants in Poland aged 60 years or older. Consequently, the number of patients with chronic diseases increases significantly with age [22]. However, the adopted type of study and the secondary epidemiological data make the unambiguous assessment of changes in the frequency of comorbidities in particular age groups of patients with sarcoidosis difficult.
Comorbid diseases generate an increase in the costs of treatment, especially in the case of sarcoidosis. The calculation of the cost of treatment is shaped by the specificities of the health care system of Poland. Our results, therefore, constitute forms of situated knowledge that need to be appraised in their socio-economic and political geographies [23, 24]. In Poland, the average cost of hospitalization for patients with interstitial lung diseases, including pulmonary sarcoidosis, is twice larger than the cost of therapy for other chronic lung diseases [12]. The important problem of the Polish system of health services should be noted: insufficient access to guaranteed services and extended waiting periods for specialized outpatient visits [25]. It cannot be excluded that this is the major reason for frequent hospitalizations of patients with sarcoidosis, which results in high medical costs. The total cost of treating patients with sarcoidosis in the USA was significantly higher than in the control group ($19,714 vs. $14,524, p<0.001) [7]. The results of our study confirmed that the average unit cost of treating patients with sarcoidosis and one coexisting disease was two times larger than the cost of the sarcoidosis itself. Unfortunately, over 80% of patients with comorbidity were hospitalized, which generates a higher unit cost of sarcoidosis treatment. Rice et al. concluded that at least 5 diseases coexisting with sarcoidosis were associated with a significantly higher cost (OR=1.7, 95%CI:1,002÷2,9, p<0,05) [6]. In the group of diseases which had caused the increasing costs of hospitalization were cardiac arrhythmia (OR=1.5; 95%CI: 1.2÷1.8, p<0.001), deficiency anaemia (OR=1.6; 95%CI: 1.2÷2.1, p<0.001), depression (OR=1.5; 95%CI:1.2÷1.9, p<0.001), diabetes (OR=1.4; 95%CI:1.2÷1.7, p<0.001), malaise and fatigue (OR=1.4; 95%CI:1.2÷1.7, p<0.001), obesity (OR=1.4; 95%CI:1.05÷1.7, p<0.05) and pulmonary aspergillosis (OR=15; 95%CI:1.2÷182.9, p<0.05) [6]. We observed that in the case in which sarcoidosis was a primary diagnosis, the average cost of treatment was twice higher in comparison to those patients in which sarcoidosis was a secondary disease.
We understand that the secondary epidemiological data collected from the registry of the National Health Fund may be the limitation of the present study. Nevertheless, the presented epidemiological picture of diseases coexisting with sarcoidosis is similar to other European data, which justifies the credibility of the results. Using official register data allowed us to estimate the occurrence of sarcoidosis, in particular, its rare clinical forms. It must be emphasized that it is not possible to estimate those values in the case of analytical research in shorter periods and smaller ranges. Similar methodological assumptions for official data are included in references [2,6,7] and discussed in this paper. Finally, the study presents an economic aspect related to the costs of treatment of sarcoidosis in Poland, and it is possible to apply direct treatment costs calculated by the National Health Fund only.
CONCLUSION
Summarizing, the standardized incidence rate of sarcoidosis has significantly decreased from the value of 13.08 (2011 year) to 7.08/100,000 (2015 year) in adult inhabitants of the Silesian Voivodship, Poland. One out of ten patients (1/10) with sarcoidosis as an underlying disease had at least one comorbid disease, in which cardiovascular (31.2%), respiratory diseases (23%), and endocrine, nutritional, and metabolic diseases (11.6%) were the most common. The frequency of coexisting diseases and demographic profile of patients with sarcoidosis were in line with other European data. The average unit costs of the sarcoidosis treatment have increased with the number of comorbidities and were related to the type of service and the basic diagnosis.
AUTHORS' CONTRITUBTION
Conceptualization, M.K. (Małgorzata Kowalska) and E.N. (Ewa Niewiadomska); methodology, E.N. (Ewa Niewiad omska); software, E.N. (Ewa Niewiadomska) and M.S. (Michał Skrzypek); formal analysis, E.N. (Ewa Niewiadomska); investigation, M.S. (Michał Skrzypek); writing—original draft preparation, E.N. (Ewa Niewiadomska).; writing—review and editing, M.K. (Małgorzata Kowalska); visualization, E.N. (Ewa Niewiadomska); supervision, M.K. (Małgorzata Kowalska). All authors have read and agreed to the published version of the manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was not any medical experiment, and the secondary character of data did not need Bioethics Committee permission. The permission of National Health Found in Katowice was obtained to use the data.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
CONFLICT OF INTEREST
The authors declare no conflict of interest. This manuscript has not been published and is not under consideration for publication elsewhere.
FUNDING
The project was funded by the Medical University of Silesia, Poland (KNW-1-209/N/8/2). This research received no external funding.
ACKNOWLEDEGMENTS
The authors would like to thank Mr. Felix Danso (Cleveland State University) for improving the English language of this manuscript.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers web site along with the published article.


