All published articles of this journal are available on ScienceDirect.
The Effectiveness of Mosquito Larva Monitoring Calendar on Knowledge, Dengue Fever Risk Behavior, and Mosquito Larva Density Rates in Malang, Indonesia
Abstract
Background
The neglected efforts of mosquito larvae observers (typically health cadres and known as Jumantik) require innovative solutions, one of which is the self-monitoring approach with the mosquito larva monitoring calendar (MLMC). This study aimed to test the effectiveness of MLMC in enhancing knowledge, reducing dengue fever (DF) risk behavior, and lowering larva densities.
Methods
A 4-month quasi-experiment was designed to evaluate the use of MLMC. The experimental group implemented MLMC, while the control group implemented the government’s basic program. Both groups were allocated randomly. Baseline and follow-up surveys in experimental and control groups were conducted, each comprising a village and housing estate. As the data was not normally distributed, the Wilcoxon signed-rank test was used to assess within-group changes, while the Student’s t-test or Mann–Whitney U test was used to compare two groups. Spearman’s test examined the relationship between MLMC utility and knowledge.
Results
Compared to control groups, there were improvements in DF risk behaviors for experimental, such as hanging clothes (p=0.008 vsp=0.130), closing the trash (p=0.027 vsp=0.004), and using mosquito nets (p< 0.001 vsp<0.001). In the follow-up survey, the MLMC received positive feedback, correlating with increased knowledge (r=0.240, p=0.006). There was no decrease in the existence of the larval stage in either the experimental or control groups (p=1.00 vsp=0.446). The control group showed reduced positive larva (p<0.001), possibly due to higher education and history of DF although not significant.
Conclusion
MLMC can reducereduce DF risk behavior, increase knowledge, and decrease mosquito larval density, contingent upon sustained compliance, assisted by Jumantik.
1. INTRODUCTION
Dengue fever (DF) is a highly transmitted vector-borne disease. It is particularly prevalent in tropical countries where Aedes aegypti is present. It was discovered in 1953 and reported in several countries such as the Philippines, Thailand, Indonesia, Myanmar, and Sri Lanka [1]. Before 1970, it was only limited to 9 countries, but by 2008, its activity has been recorded in 69 countries from the WHO regions of Southeast Asia, Western Pacific, and the Americas, with Asia accounting for over 70% of the worldwide disease cases [2]. Brady et al.’s study reported that about 128 countries were affected by dengue by 2012 [3].
Indonesia has the most cases of DF among Southeast Asia countries from 1985 to 2009 [1]. In 2016, it reported 204,171 cases and 1,589 deaths, indicating a significant increase compared to 129,650 in 2015 [4]. This trend was also observed in Malang City, a city in East Java, Indonesia. The city experienced three notable increases in DF cases since 2014, specifically in 2015, 2016, and 2019. There were 187, 298 (3 deaths), and 464 (3 deaths) in 2014, 2015, and 2016, respectively. The prevalence of this disease in Sawojajar Urban Village, within Malang City, showed a decrease in 2017 with 105 cases that further decreased to 82 in 2018 [5]. However, in 2019, the disease increased to 300 cases [6]. An entomological survey indicated that 96.3% of the mosquito larvae in Kedung Kandang Sub-district were Aedes aegypti, while the remaining belong to the Culex and Anopheles species [7].
Despite a direct relationship not being established between the vector index and dengue transmission, vector control measures serve as a strategy to combat DF transmission [8]. Therefore, it is important to control Aedes aegypti mosquito in order to prevent DF transmission.
Successful household vector control relies on effective coordination among community members, government, and the private sector. Improved management of containers as a larval habitats, is needed due to the high house index [9]. Community-based larval habitat observers, known as Juru Pemantau Jentik (Jumantik), aim to promote behavioral changes for a cleaner environment. However, they sometimes encounter resistance from the community, which can hinder their efforts.
Jumantik, are health cadres who actively remove mosquito breeding sites and inspect for Aedes aegypti mosquito larvae [10]. A study reported that 1% of the community members denied researcher access to inspect potential larval habitats in their homes [7]. This issue was supported by Jumantik during a Focus Group Discussion
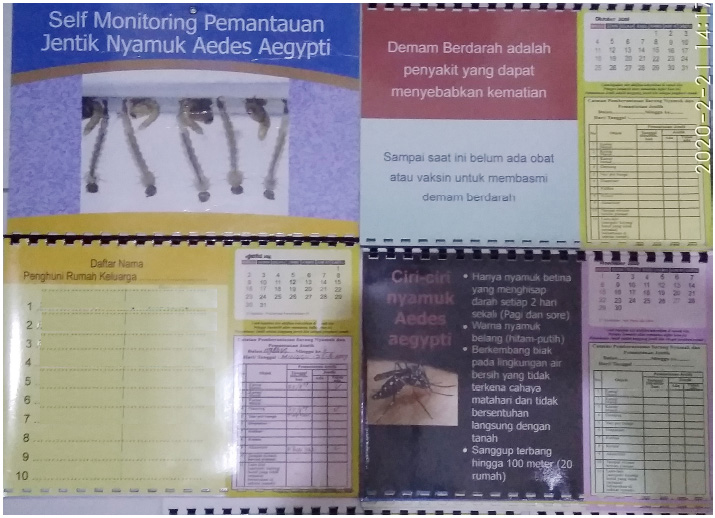
The mosquito larva monitoring calendar (MLMC).
(FGD). As a solution, self-monitoring method was proposed, involving a mosquito larva monitoring calendar (MLMC) or Kalender Pemantauan Jentik (KPJ), which was . suggested by a community leader during the FGD [11].
The Mosquito Larva Monitoring Calendar (MLMC) provides images and information related to dengue fever (DF) as Fig. (1), featuring a checklist for community members to independently document their cleaning efforts in potential larva habitats. The self-monitoring approach encourages individuals to report their activities. A trial showed that using mosquito nets improved control [12].
The MLMC model aims to enhance DF knowledge, promote better behaviors, and reduce mosquito larvae densities at home. Therefore, this study aimed to assess the effectiveness of applying self-monitoring model using MLMC in increasing knowledge, decreasing DF risk behavior, and lowering larva densities.
2. MATERIALS AND METHODS
2.1. Study Design
This study used a quasi-experimental design or community intervention known as controlled before and after (CBA), specifically the pre-post with control design [13]. The intervention involved implementing self-monitoring model using MLMC for 4 months, spanning from October 2009 to January 2010. This was conducted in selected households of a neighborhood (Rukun Warga–RW) located in Sawojajar Urban Village, Kedung Kandang Sub-District, Malang City, Indonesia. The village covers an area of 18,125 hectares and is situated at an elevation of 250 m above sea level. It receives an average annual rainfall of 1280 mm, and the topography of the area is mostly flat to choppy, comprising 90% of the land, with an average air temperature ranging from 24°C to 31°C. Furthermore, the village consists of 118 RT (Rukun Tetangga–Sub-RW) and 16 RW, with a population density of about 4,066 inhabitants/km2. Most residents rely on the local water company as a primary source for their daily needs [14]. This study has received approval for all the procedures conducted in this research.
The prevalence of DF peaked in July 2009, when 134 cases were reported, up from 129 in April 2009, according to statistics from the Malang City Health Office [15]. In addition, DF cases were higher in the first month of 2009 than in the same month in 2008. These statistics highlight the urgent need for intervention to prevent further transmission of the disease. Among the urban villages in Malang City, Sawojajar Urban had the highest dengue cases during that period.
This study involved the control and experimental groups. The control group consisted of a neighborhood (RW) that did not apply self-monitoring model, with one cluster RW from the village and another from the housing estate. The experimental group comprised community members within a neighborhood that applied self-monitoring model, also with one cluster RW from the village and another from the housing estate. Certain criteria were considered in selecting the RW, such as the highest number of dengue cases reported in the last 6 months, the lowest larva-free rate, and the high level of activity of cadres. Four of the 16 RWs assessed met the criteria, 2 of which were village-type and 2 were housing estate-type. Furthermore, two RWs with village types were randomly and equally divided into the control and experimental groups. Similarly, the two RW types of housing estate were randomly and equally divided into groups. The division of control groups and treatment groups as well as research workflow and data analysis were depicted in Fig. (2). In 2009, data from the Community Health Center reported that there were 22 and 2 dengue cases in the experimental and control groups, respectively.
The study sample size was determined using a formula appropriate for a limited population. A previous study [16] reported that the proportion of larval positive houses were 0.39. We used Power and Sample Size Program Software to calculate the sample size. By using α =0.05, change in proportion=0.16, power=80%, d=0.05 we get the number of samples per group is 148. Then we round it up to 150 per group. Furthermore, the number of samples was rounded up to 300 houses and divided equally into the control and experimental groups. The respondents in the study were household members who were capable of providing information about their houses related to DF. The criteria for selecting households for the experimental group included their willingness to apply the calendar, while that for the control group involved those with similar characteristics in terms of education to the experimental group.
2.2. Data Collection
Before obtaining consent from the participants, the researchers provided information about DF and its prevention to both groups. While, detailed information about the objectives of the study, the utility of MLMC, and methods of filling it out to all of the RTs were given to the experiment group only in their monthly women’s meeting. All participants were adults and gave full written informed consent.in this study. All participant rights were protected; even the interventions given are not absolute obligations. Therefore, we considered the percentage of MLMC filling. The participants were instructed to fill out MLMC once a week, after which 50% of the sample was monitored in terms of the implementation of MLMC and inspected for the existence of mosquito larvae by jumantiks. Twice a week, research assistants monitor the activities of jumantiks and survey 20% of the sample houses for the implementation of MLMC and inspected for the existence of mosquito larvae. Furthermore, once a month, the researchers monitored the jumantiks and surveyed 10% of the sample houses. The jumantiks were provided an incentive to enhance their participation. Baseline and follow-up surveys were conducted to examine whether the intervention affected behavioral change
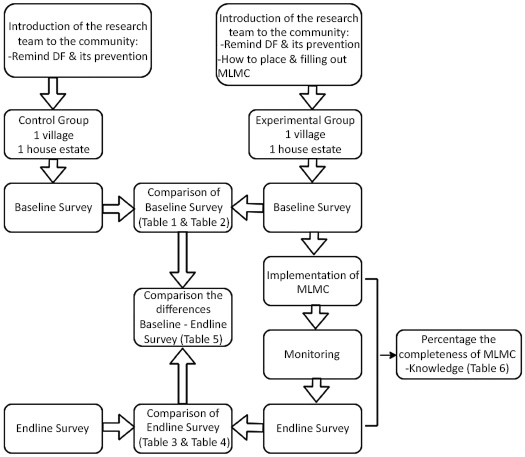
The division of control groups and treatment groups as well as research workflow and data analysis.
related to DF risks, knowledge about DF, and larva density rate. A larva density survey was conducted by observing the mosquito larvae directly in 8 types of containers. The observers were equipped with flashlights. In the questionnaire, the questions about behavior used a Likert scale with the code 1 = often, 2 = always, 3 = sometimes, 4 = occasionally, 5 = never for positive questions, while for negative questions the opposite code was used. For questions about knowledge, the Guttman scale was used. The questionnaire has been tested for its validity and reliability.
2.3. Data Processing
This study analyzed sociodemographic factors such as the education level of respondents, the highest level of education of family members, total gross income of all family members, respondent age, the age of the youngest family member, occupant load density, and history of DF among family member in both groups. This study was conducted to determine the similarity of initial conditions. The independent variable was the application of self-monitoring model. The dependent variables were knowledge about DF, DF risk and preventive behavior, the presence of larvae at home, and larva density rate. DF risk behaviors consist of three negative statements that assessed several habits, such as sleeping in the morning (08.00–12.00), sleeping in the afternoon (15.00–18.00), and hanging clothes in the room (not a wardrobe). Meanwhile, positive statements include cleaning the tub at least once every week, littering responsibly, closing the clay barrel and trash can, utilizing mosquito nets, and applying insect repellent or anti-mosquito lotion. Knowledge of the disease consists of 19 questions, such as an understanding of DF, characteristics of mosquitoes, symptoms of DF, first aid, and DF prevention slogans of Draining (Menguras) the tub, Closing (Menutup) containers, and Burying (Mengubur) garbage. The slogan became known as 3M.
2.4. Statistical Analysis
The independent t-test was performed to compare the characteristics of age between the control and experimental groups; the Mann–Whitney U test to compare other characteristics, and DF risk behavior between the groups; and the Wilcoxon test to compare behavior scores before and after the intervention. To analyze the presence of larvae and the history of DF among family members, the Chi-square test was conducted using https://www.openepi.com/TwobyTwo/TwobyTwo.htm. This test compared the variables between the control and experimental groups, as well as before and after treatment. Microsoft Excel was used to run Spearman’s correlation test (since the data is not normally distributed) which analyzed the impact of completeness of filling out MLMC forms on knowledge. It was important to note that the analysis did not differentiate between village and housing estate types because the unit of analysis is the individual.
3. RESULTS
3.1. Baseline Survey
Primary data were gathered from 300 respondents. The oldest respondents were 82 years old, the youngest were 15 years old, and the average age was 43.91. Most of the respondents (86.3%) were women, while senior high school was the most common last educational level (40.3%). Table 1 shows a comparison of the sample characteristics before the intervention. It suggests that both groups share the same sociodemographic characteristics. Similar to behavior scores, larva density, and family history of dengue did not vary between the groups.
3.2. Follow-up Survey
The age range of respondents in the follow-up survey was 18–82, with a mean age of 43.5 years. Most of the respondents (254 or 84.7%) were women, while senior high school (130–43.5%) made up the highest educational level in the families. The sample characteristics for the follow-up survey were largely the same, as shown in Table 2. However, there was a significant difference in the greatest level of education (high school to university) between the experimental and control groups. Table 2 also shows that there were more homes having a history of dengue in the control group than in the experimental group. Despite living in the same home, the respondents in the follow-up survey are not always the same as those in the baseline survey, which accounts for the change in educational attainment. Included in this are the challenges encountered in keeping consistent respondents before and after the intervention.
3.3. Impact on Knowledge
In the experimental group, 13 (10.2%) of the 150 households filled it completely as instructed. Table 3 shows the performance of MLMC from the perspective of the respondents. The Spearman’s correlation test showed that the percentage of filling MLMC was significantly related to the knowledge about DF. However, the relationship was weak with r = 0.240 (p = 0.006). These indicated a direct proportionality between the percentage of completeness in filling out MLMC and the score of knowledge of the users.
| S.No. | Variable |
Experimental Group (n=150 houses) |
Control Group (n=150 houses) |
p-value |
|---|---|---|---|---|
| (Mean± SD) | (Mean± SD) | |||
| 1 | Average age | 45.02 ± 13.76 | 42.68 ± 12.09 | 0.140† |
| 2 | Average total gross income of all family members (in million) | 1.93 ± 0.93 | 2.03 ± 0.95 | 0.380 |
| 3 | Average occupant load density | 0.05 ± 0.03 | 0.05 ± 0.03 | 0.950 |
| 4 | Average behavior score | 3.84 ± 0.44 | 3.83 ± 0.58 | 0.670 |
| - | - | n (Percentage) | n (Percentage) | - |
| 5 | The highest education (High School - University) of respondents | 81 (54.0) | 96 (64.0) | 0.100δ |
| 6 | The highest education level (High School - University) of family members | 133 (88.7) | 143 (95.3) | 0.060δ |
| 7 | History DF among Family member | - | - | 1.000δ |
| - | No | 132 | 132 | - |
| - | Yes | 18 | 18 | - |
| 8 | Sex | - | - | - |
| - | Male | 24 (58.5) | 17 (41.5) | 0.313 |
| - | Female | 126 (48.6) | 133 (51.4) | - |
| 9 | Existence of larvae (House Index) | 30 (20.0) | 29 (19.3) | - |
| S.No. | Variable |
Experimental Group (n=150 houses) |
Control Group (n=150 houses) |
p-value |
|---|---|---|---|---|
| (Mean±SD) | (Mean±SD) | |||
| 1 | Average age | 44.96 ± 13.51 | 42.77 ± 11.60 | 0.330† |
| 2 | Average total gross income of all family members (in million) | 1.9 ± 0.98 | 1.87 ± 0.84 | 0.460 |
| 3 | Average occupant load density | 0.06 ± 0.06 | 0.05 ± 0.04 | 0.600 |
| 4 | Average behavior score | 3.82 ± 0.51 | 3.96 ± 0.46 | 0.030 |
| - | - |
n (Percentage) |
n (Percentage) | - |
| 5 | The highest education (High School - University) of respondents | 94 (62.7) | 107 (71.8) | <0.001 δ |
| 6 | The highest education level (High School – University) of family members | 130 (86.7) | 139 (93.3) | 0.130 δ |
| 7 | History DF among Family member | - | - | 0.180 δ |
| - | No | 127 | 117 | - |
| - | Yes | 23 | 33 | - |
| 8 | Sex | - | - | 0.037 |
| - | Male | 16 (34.8) | 30 (65.2) | - |
| - | Female | 134 (52.8) | 120 (47.2) | - |
| 9 | Existence of larvae (House Index) | 29 (19.7) | 23 (15.3) | - |
| S.No. | Respondents’ Opinions | Strongly Agree | Agree | Neutral | Disagree | Strongly Disagree | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| - | - | n | % | n | % | n | % | n | % | n | % | n | % |
| 1 | Easy filling | 44 | 34.4 | 59 | 46.1 | 12 | 9.4 | 11 | 8.6 | 2 | 1.6 | 128 | 100 |
| 2 | Complete information or messages | 33 | 25.8 | 73 | 57.0 | 17 | 13.3 | 5 | 3.9 | 0 | 0 | 128 | 100 |
| 3 | Attractive pictures | 38 | 29.7 | 76 | 59.4 | 12 | 9.4 | 2 | 1.6 | 0 | 0 | 138 | 100 |
| 4 | Attractive color | 39 | 30.5 | 75 | 58.6 | 12 | 9.4 | 2 | 1.6 | 0 | 0 | 128 | 100 |
| 5 | Easy-to-read font types and sizes | 27 | 21.1 | 76 | 59.4 | 11 | 8.6 | 13 | 10.2 | 1 | 0.8 | 128 | 100 |
| 6 | Interesting design | 32 | 25.0 | 76 | 59.4 | 14 | 10.9 | 5 | 3.9 | 1 | 0.8 | 128 | 100 |
| 7 | Design/size suitable for home use | 27 | 21.1 | 82 | 64.1 | 9 | 7.0 | 9 | 7.0 | 1 | 0.8 | 128 | 100 |
| 8 | Very needed | 37 | 28.9 | 65 | 50.8 | 18 | 14.1 | 6 | 4.7 | 2 | 1.6 | 128 | 100 |
3.4. Impact on Behavior
Changes in behavior score were not significant in both groups. However, after the intervention, the experimental group showed improvement in certain behavior such as hanging clothes in the room (not the wardrobe), closing the trash can, and using mosquito nets. Meanwhile, control group showed improvements in morning naps, closing the trash can, and using mosquito nets (Table 4).
3.5. Impact on the Existence of Larval-stage Mosquitoes
Based on observations of 300 houses in the baseline larva survey, larval-stage mosquitoes were discovered in 59 houses (39.3%). It is important to note that some households declined inspections on several objects, while others had no objects to be inspected (Table 5). The most common location for larval-stage mosquitoes in both groups was the bathroom, followed by the refrigerator wastewater container, clay barrels, water dispensers, flowerpots, and other containers.
In the follow-up survey, larval-stage mosquitoes were discovered in 52 (35.0%) out of 300 households (Table 6). As observed, the bathroom remained the primary location with the most larval-stage mosquitoes in the two groups. It was followed by containers such as refrigerator wastewater containers, other containers, clay barrels, and flowerpots. There was no significant difference in the presence of larvae before and after the intervention in both groups. However, there was a slight decline in the house index.
3.6. Monitoring the Implementation of Interventions
According to data gathered from 103 homes that researchers monitored, 90.3% of the homes reported that jumantiks had visited them during the study, 68.9% of the MLMC were stuck on the wall, 65% of the MLMC were fully visible, 22.3% of the MLMC had mistaken in their filling out, and 25.2% of the homes discovered larvae. The study also suggests that households use their MLMC more effectively while conducting this monitoring activity. The results of jumantiks performance monitoring showed that there was consistency in monitoring data on the presence of larvae between researchers and jumantiks, with a Kappa value of 0.589 (p < 0.001).
| Variable | Control Group (Before vs. After Intervention) | Experimental Group (Before vs. After Intervention) | ||
|---|---|---|---|---|
| Mean | p-value | Mean | p-value | |
| Morning naps (08.00 - 12.00) | 3.78 Vs 4.09 | 0.038* | 4.10 Vs 3.88 | 0.600 |
| Afternoon naps (15.00 - 18.00) | 3.97 Vs 3.87 | 0.271 | 3.77 Vs 3.94 | 0.558 |
| Hanging clothes in the room (not the wardrobe) | 2.47 Vs 3.09 | 0.130 | 2.67 Vs 3.29 | 0.008* |
| Cleaning the tub at least once a week | 4.63 Vs 4.52 | 0.002** | 4.80 Vs 4.50 | 0.016** |
| Littering well | 4.59 Vs 4.35 | 0.298 | 4.87 Vs 4.88 | 0.176 |
| Closing the trash can | 3.16 Vs 4.09 | 0.004* | 3.23 Vs 3.65 | 0.027* |
| Covering the clay barrel | 4.69 Vs 4.39 | <0.001** | 4.87 Vs 4.56 | <0.001** |
| Using mosquito net | 2.69 Vs 3.30 | <0.001* | 2.23 Vs 2.53 | <0.001* |
| Using other methods*** | 3.13 Vs 3.57 | 0.280 | 3.33 Vs 2.62 | 0.009** |
| Behavior | 3.83 ± 0.58 Vs 3.96 ± 0.46 | 0.142 | 3.82 ± 0.44 Vs 3.82 ± 0.51 | 0.096 |
| Existence of larval-stage mosquito† | 29 (19.3) Vs 23 (15.3) | 0.446 | 30 (20.0) Vs 29 (19.7) | 1.000 |
| S.No. | Objects |
Experimental Group (n=150 houses) |
Control Group (n=150 houses) |
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (+) | - | (-) | - | NA | - | NH | - | (+) | - | (-) | - | NA | - | NH | - | ||
| n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | ||
| 1 | Bathroom | 18 | 12.0 | 127 | 84.7 | 2 | 1.3 | 3 | 2.0 | 23 | 15.3 | 122 | 81.3 | 3 | 2.0 | 2 | 1.3 |
| 2 | Water dispenser | 3 | 2.0 | 44 | 29.3 | 1 | 0.7 | 102 | 68.0 | 1 | 0.7 | 33 | 22.0 | 0 | 0.0 | 116 | 77.3 |
| 3 | Refrigerator wastewater container | 1 | 0.7 | 84 | 56.0 | 3 | 2.0 | 62 | 41.3 | 6 | 4.0 | 81 | 54.0 | 3 | 2.0 | 60 | 40.0 |
| 4 | Flower vase/flowerpot | 2 | 1.3 | 28 | 18.7 | 0 | 0.0 | 120 | 80.0 | 0 | 0.0 | 28 | 18.7 | 0 | 0.0 | 122 | 81.3 |
| 5 | Pool/aquarium | 1 | 0.7 | 26 | 17.3 | 1 | 0.7 | 122 | 81.3 | 0 | 0.0 | 23 | 15.3 | 0 | 0.0 | 127 | 84.7 |
| 6 | Clay barrel | 4 | 2.7 | 66 | 44.0 | 1 | 0.7 | 79 | 52.7 | 1 | 0.7 | 54 | 36.0 | 2 | 1.3 | 93 | 62.0 |
| 7 | Pet water dispenser | 0 | 0.0 | 18 | 12.0 | 2 | 1.3 | 130 | 86.7 | 0 | 0.0 | 17 | 11.3 | 1 | 0.7 | 132 | 88.0 |
| 8 | Others* | 1 | 0.7 | 39 | 26.0 | 0 | 0.0 | 110 | 73.3 | 0 | 0.0 | 36 | 24.0 | 0 | 0.0 | 114 | 76.0 |
| S.No. | Objects |
Experimental Group (n=150 houses) |
Control Group(n=150 houses) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (+) | - | (-) | - | NA | - | NH | - | (+) | - | (-) | - | NA | - | NH | - | ||
| n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | ||
| 1 | Bathroom | 23 | 15.3 | 121 | 80.7 | 5 | 3.3 | 1 | 0.7 | 17 | 11.3 | 124 | 82.7 | 7 | 4.7 | 2 | 1.3 |
| 2 | Water dispenser | 1 | 0.7 | 40 | 26.7 | 1 | 0.7 | 108 | 72.0 | 1 | 0.7 | 46 | 30.7 | 2 | 1.3 | 101 | 67.3 |
| 3 | Refrigerator wastewater container | 2 | 1.3 | 67 | 44.7 | 1 | 0.7 | 80 | 53.3 | 9 | 6.0 | 76 | 50.7 | 13 | 8.7 | 52 | 34.7 |
| 4 | Flower vase/flowerpot | 2 | 1.3 | 10 | 6.7 | 0 | 0.0 | 138 | 92.0 | 0 | 0.0 | 12 | 8.0 | 0 | 0.0 | 138 | 92.0 |
| 5 | Pool/aquarium | 1 | 0.7 | 12 | 8.0 | 0 | 0.0 | 137 | 91.3 | 0 | 0.0 | 23 | 15.3 | 2 | 1.3 | 125 | 83.3 |
| 6 | Clay barrel | 2 | 1.3 | 71 | 47.3 | 1 | 0.7 | 76 | 50.7 | 1 | 0.7 | 43 | 28.7 | 2 | 1.3 | 104 | 69.3 |
| 7 | Pet water dispenser | 0 | 0.0 | 22 | 14.7 | 1 | 0.7 | 127 | 84.7 | 0 | 0.0 | 23 | 15.3 | 1 | .7 | 126 | 84.0 |
| 8 | Others* | 1 | 0.7 | 21 | 14.0 | 0 | 0.0 | 128 | 85.3 | 4 | 2.7 | 25 | 16.7 | 2 | 1.3 | 119 | 79.3 |
4. DISCUSSION
At the beginning of the study, both the experimental and control groups had similar sociodemographic characteristics. However, due to the difficulty in maintaining the same individuals for the follow-up survey, there were slight differences in characteristics in terms of sex, respondents’ education, and the average age of the youngest family members between the two groups. Specifically, the control group showed better characteristics in terms of respondents’ education, more men, and the average age of the youngest family members than the experimental group. The main result of this study found no significant changes in behavior for the experimental group. On the other hand, beneficial changes occur in certain behavioral factors. A study conducted in Malaysia reported that education increased dengue prevention behavior. In addition, it was observed that the men showed slightly better behavior improvement than the women [17].
Based on the responses provided by the respondents, the control group in the follow-up survey exhibited a higher history of dengue cases than the experimental group. This affects the alertness in responding to and preventing dengue episodes from reoccurring. In contrast, it should be anticipated that areas with low case numbers may experience an increase in cases in the next period. A study from Malaysia reported that a history of this disease enhances its preventive behavior [18] [19]. However, studies conducted in Peninsular Malaysian regions [17] and Karachi [20] showed no association between history of dengue and preventive behaviors.
Despite the intervention, the presence of larvae did not reduce in the experiment group, while the control group showed slightly better results. The baseline and follow-up survey confirmed that the bathroom was the main larva habitat and this was consistent with a previous study conducted in Malang Raya [7]. Several families have multiple bathrooms, some of which were not accessible for observation due to privacy concerns. The intervention employed could not significantly decrease larva density. This was consistent with a study conducted in Yogyakarta on the use of Larvae Control Card. There was no significant difference in larva-positive containers and houses after the use of Larvae Control Card [21].
In addition, not all of the households in the experiment group were willing to receive and fill out MLMC completely. Among 150 houses, only 128 households were willing to receive the calendar. Meanwhile, 13 (10.2%) out of 150 households filled out the MLMC completely. This was smaller compared to the study conducted in Yogyakarta’s study, where the participation rate was 28.3% [21]. Only a few respondents completed the filling process, despite most of the respondents giving the MLMC positive feedback (Table 4), such as easy filling, complete information, appealing photos and color, easy reading, engaging and suitable design, and extremely required. This limited compliance might explain the insignificant decrease in larva density observed in this study. Although this research shows that compliance with filling out the MLMC is positively related to increased knowledge. It may take longer for some people to get into the habit of immediately writing down their larva observation activities at home to MLMC. According to Everett Rogers’ theory, the success of an initiative requires the involvement of five community groups: innovators, early adopters, early majority, late majority, and laggard [22]. Jumantiks, as collaborators conducting regular visits and monitoring the behavior of community members, could serve as intermediary agents to support the success of health centers’ programs [23]. In this case, jumantiks can be fully involved in supervising the filling of the MLMC for self-monitoring or independent surveys by residents.
Surveys are a valuable method to measure health behavior in the community. They play a crucial role in program planning and program evaluation [24]. Conducting larval-stage monitoring and recording the results independently using MLMC may be new activities for the community. However, people are not familiar with recording the results. Therefore, adopting this intervention technique requires several steps, such as activating change, implementing specific information areas of education and self-monitoring, developing skills, obtaining environmental resources, and building social support [25].
Sometimes, the results can also overrepresent actual conditions, as respondents may claim to have taken several preventive actions when they have only performed one. Despite this concern, when implemented correctly, self-monitoring can serve as a reminder and encouragement for the community to improve their behavior. In this study, MLMC acts as an educational medium and monitoring tool to drive behavior change within the community. The results were in line with the study conducted in Bandung, which also used calendars as an education and monitoring tool [23]. Similarly, a study conducted in Cuba revealed that a community-based environmental management approach outperformed conventional methods such as entomological surveillance, killing adult mosquitoes, destroying mosquito nests, and health education [26].
The MLMC did not alter communal behavior, but it did offer some promise when we incorporated households and cadres. However, there is still room to increase the breadth and depth of community involvement. In this case, the role of the community becomes very important. Involving and mobilizing communities, improving vector surveillance, and monitoring and evaluating interventions are three of the four pillars of the global vector control response 2017–2030 to achieve effective, locally relevant, and long-term vector control. The other pillar is fostering cross- and intra-sectoral cooperation [27].
Improving community behavior plays a crucial role in integrated vector management (IVM), a strategic approach to vector control developed by the WHO. One of the key elements that need to be emphasized in this case is capacity building for the community [28]. Religious leaders, community leaders, and health centers play a significant role in building cooperation among community members to fight DF, as evidenced in Pakistan [29]. For the urban community, a combination of bottom-up and top-down approaches is essential to effectively reach settlements and public facilities legally and inclusively [30]. The involvement of the community is of utmost importance, and models aimed at increasing participation in dengue prevention remain relevant in light of social, cultural, and technological developments. With the development of technology and the capabilities of the community, self-monitoring for online mosquito larvae surveillance, which is supervised by jumantik and puskesmas (Community Health Center), needs to be considered for use.
This study has some limitations. Assessing knowledge and practices before and after intervention among residents, with the same person, can be challenging due to high mobility. However, involving respondents from the same household is expected to show similar performance.
CONCLUSION
The Self-Monitoring Model with MLMC could effectively reduce some DF risk behavior, enhance knowledge if associated with its completion, and decrease mosquito larva density even if not significantly. However, implementing the self-monitoring model in the community using MLMC needs commitment. This model requires the role of Jumantik and Puskesmas to monitor the implementation by community members. Future studies should focus on exploring the history of dengue in the family. Moreover, a combination of community engagement as in IVM and the global vector control response with the self-monitoring model is expected to obtain more significant changes in knowledge, behavior, and larva density. Furthermore, the use of online surveys needs to be considered in the next trial.
AUTHORS' CONTRIBUTION
L.Z.: Drafted the manuscript, designed the study, validation, conducted data collection, and analyzed the findings; L.E.F.: Drafted the manuscript, designed the study, reviewed and edited the manuscript; H.R.: designed the study, conducted data collection, and analyzed the findings. All authors have reviewed and approved the final manuscript.
LIST OF ABBREVIATIONS
| DF | = Dengue Fever |
| MLMC | = Mosquito Larva Monitoring Calendar |
| KPJ | = Kalender Pemantauan Jentik (MLMC in Indonesian) |
| IVM | = Integrated Vector Management |
| RT | = Rukun Tetangga (a cluster of houses, usually around 50 houses) |
| RW | = Rukun Warga (a cluster of RT, usually more than 1 RT, until 20 RT) |
| CBA | = Controlled Before and After |
| DENV | = Dengue Virus |
| FGD | = Focus Group Discussion |
| 3M | = Menguras Mengubur dan Menutup (activity of Draining, Closing, and Burying in Dengue Prevention). |
ETHICS APPROVAL AND CONSENT TO PARTI-CIPATE
All ethical standards were strictly followed and the identity and confidentiality of respondents' data were ensured. The Ethics Committee of the Faculty of Medicine at Universitas Brawijaya, Indonesia authorised our research report after thoroughly reviewing it to verify that it was ethically appropriate.( certificate no. 70/UNIO.F08.10/PN/2023).
HUMAN AND ANIMAL RIGHTS
All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
The source of data and materials mentioned in the manuscript are available within the article.
FUNDING
This study was funded by the Directorate General of Higher Education, Ministry of Education and Culture of Indonesia, Indonesia Awards/Grant number: 531/D3/PL/2009; 20 May, 2009.
ACKNOWLEDGEMENTS
The authors would like to thank the Health Office Malang City, Puskesmas Officers, and health cadres for allowing this research to be carried out. We would also like to thank the study participants for their time and participation.


