All published articles of this journal are available on ScienceDirect.
Correlation Between Gestational Age by Last Menstrual Period (LMP) and Fetal Biometry Using Ultrasonography in the First Trimester in Ethnic Minangkabau
Abstract
Introduction
Ultrasonography (USG) is a reliable method for measuring fetal biometry and estimating birth weight.
Methods
This cross-sectional study examines fetal growth biometry in the Minangkabau ethnic group, with data collected from pregnant women who attended prenatal check-ups at the Fetomaternal clinic of DR. M. Djamil General Hospital, Padang, in December 2020. A Pearson correlation test was used for bivariate analysis due to the normal distribution of the data (p value <0.05), followed by multivariate analysis using linear regression.
Results
Ultrasound examinations were conducted on 400 pregnant women in their first trimester (4-13 weeks) for prenatal assessments. Pearson correlation analysis demonstrated a strong relationship (r=0.94, p=0.0001) between crown-rump length (CRL) and gestational age (GA) as determined by the last menstrual period (LMP).
Conclusion
We have established a national standard for fetal growth assessment and first-trimester ultrasound pregnancy dating in Indonesia, specifically designed for the Minangkabau ethnic population.
1. INTRODUCTION
The evaluation of fetal growth plays a pivotal role in predicting newborn health and the likelihood of perinatal morbidity and mortality [1]. Studies show that fetal size and birth weight are notably affected by the race and ethnicity of the mother [2, 3]. For instance, one study reported that babies of European descent were, on average, 225.5 grams heavier than those of Chinese descent and 254.6 grams heavier than infants of South Asian descent. [2] This pattern, which emerges early in development, points to genetic influences or maternal physiological factors as the main contributors rather than lifestyle or dietary habits in later life stages [2, 4, 5].
Since the discovery of ultrasonography (USG) technology over 40 years ago, fetal biometry using ultrasound has become the gold standard and is regarded as a reliable method for estimating birth weight [6, 7]. Advances in science have introduced various approaches for estimating fetal weight, such as the formulas developed by Jeanty, Aoki, Campbell, Shepard, and Hadlock. Recent studies continue to validate and refine these approaches. For example, Mascherpa (2024) highlighted that while different charts and definitions for fetal growth restriction exist, they do not significantly improve the prediction of adverse outcomes, indicating the ongoing need for precise and population-specific growth charts [8]. Furthermore, Dagklis (2024) reaffirmed the accuracy of Hadlock's formula in estimating fetal weight, emphasizing its continued relevance in clinical practice [9]. Moreover, new research methods are still evolving [10]. More than two decades ago, Hadlock introduced formulas and growth charts, and Pressman et al. found that the Hadlock formula (GS, CRL, BPD, HC, AC, HC, FL) was more accurate than others in estimating fetal weight in 138 pregnant women [11-14].
Longitudinal research projects, such as Intergrowth-21 (IN-21), have established fetal biometric guidelines applicable to various population groups. Studies from the National Institute of Child Health and Human Development (NICHD) and the WHO have also contributed significantly to reevaluating fetal growth standards [12]. However, as noted by Guellec et al., these multinational studies often represent only a fraction of the global population, necessitating the development of growth charts that are tailored to specific demographic and environmental contexts [15]. These tailored charts must account for factors such as high-altitude environments, regional climatic extremes, economic status, healthcare access, and cultural differences, which can significantly influence fetal growth patterns. Zazara et al. also emphasized the importance of considering environmental factors in fetal growth assessments, particularly in relation to respiratory health outcomes [16]. Moreover, the work of Chen et al. indicated that maternal factors, such as pre-pregnancy body mass index, can also impact fetal growth trajectories, further complicating the establishment of universal growth standards [17]. This highlights the necessity for localized growth charts that reflect the unique characteristics of different populations, as these factors can lead to significant variations in fetal development and birth weight outcomes.
The role of ethnicity in fetal growth is vital, making it problematic to apply biometric reference ranges from uniform populations to diverse or heterogeneous ones. For instance, a study in the United States on singleton pregnancies ranging from 17 to 22.9 weeks found that African-American fetuses had a smaller abdominal circumference (AC) when compared to Caucasian fetuses. Because AC is a significant predictor of estimated fetal weight, this could result in underestimating the weight of African-American fetuses [18]. In Sudan, new reference charts and equations have been introduced for key measurements such as biparietal diameter, head circumference, abdominal circumference, femur length, and fetal weight, specifically designed for the Sudanese population [19].
Indonesia currently lacks a standardized reference for fetal weight percentiles. As a developing nation with a population exceeding 200 million and rich ethnic and cultural diversity, it is important to establish an appropriate standard reference to prevent mislabeling or mismanaging pregnancies, whether involving normal fetuses or those needing additional care [12].
The establishment of a standard reference for fetal growth within the Indonesian population, particularly focusing on the Minangkabau ethnic group, is crucial for understanding and improving maternal and neonatal health outcomes. This study is particularly significant given the diverse maternal demographics, including variations in age and socioeconomic status, which are known to influence fetal growth patterns.
Research indicates that fetal growth standards should be tailored to specific populations due to the biological variability that exists across different ethnic groups. For instance, Fattah et al. demonstrated that the Indonesian fetal growth standard provides a more accurate predictive ability for low Apgar scores in small for gestational age (SGA) neonates compared to international standards like Hadlock's [20]. This finding underscores the necessity of developing localized growth charts that reflect the unique characteristics of the population being studied. Moreover, Cheng et al. emphasized the importance of assessing the suitability of international fetal growth references, as fetal growth is influenced by both genetic and environmental factors that can vary significantly between populations [21]. The need for customized growth charts is further supported by Maso et al., who argue that specific population-based antenatal references are essential for accurately assessing fetal growth, especially in settings where resources are limited [22]. This is particularly relevant in Indonesia, where healthcare access and quality can differ widely across regions.
The implications of maternal demographics on fetal growth cannot be overstated. Studies have shown that maternal age, nutritional status, and socioeconomic background play critical roles in determining fetal growth trajectories. For instance, the use of cross-sectional growth charts has been identified as a primary screening tool for assessing fetal growth, with customization improving the detection of high-risk fetuses [23]. This suggests that incorporating diverse maternal demographics into the fetal growth standard development process is vital for enhancing its applicability and effectiveness. Furthermore, the work of Grantz et al. highlights that universal fetal growth standards may not adequately capture the nuances of growth patterns in different populations due to the interplay of environmental and genetic factors [24]. This reinforces the argument for developing localized standards that consider the specific characteristics of the Minangkabau ethnic group, which may differ from those of other populations.
This study sought to establish a standard reference for fetal growth in the Indonesian population, specifically focusing on the Minangkabau ethnic group. Additionally, the researchers aimed to explore the relationship between various ultrasound parameters (GS, CRL, BPD, and FL) and gestational age based on the last menstrual period (LMP). The findings from this study are expected to offer more comprehensive parameters and improved accuracy in estimating fetal growth [11-14].
2. METHODS
This analytical investigation employed a cross-sectional approach to examine fetal growth biometry within the Minangkabau ethnic group. The study took place in December 2020 at the Fetomaternal Clinic of M. Djamil Hospital in Padang, focusing on pregnant women who visited the clinic and satisfied the specified inclusion and exclusion criteria. To be included, participants had to provide informed consent, have a singleton intrauterine pregnancy, be between 28 and 40 weeks of gestation, and possess a normal body mass index (BMI of 18.5 to 24.9). Those excluded from the study were pregnant individuals with fetal abnormalities or a history of medical conditions such as myomas, cysts, hypertension, diabetes, kidney or heart diseases, autoimmune disorders (e.g., SLE), as well as those who smoked, consumed alcohol or drugs, or used long-term medications like steroids or antihypertensives. In total, 400 samples were gathered for gestational ages ranging from 4 to 13 weeks.
The sampling method used in this study was a consecutive sampling approach, targeting all pregnant women who attended the Fetomaternal Clinic at DR. M. Djamil General Hospital in Padang and met the specified inclusion criteria. There were no participants who fell under the exclusion criteria. Various fetal biometry parameters, including gestational sac (GS) and crown-rump length (CRL) measured in centimeters, as well as gestational age (GA) measured in weeks, were documented. A statistical analysis was carried out to outline the characteristics of both dependent and independent variables, with findings displayed as frequency distributions in tabular form. For normally distributed data, bivariate analysis was performed using the Pearson correlation test, and a p-value of <0.05 was regarded as statistically significant. Additionally, multivariate analysis was conducted with linear regression to evaluate variables with p-values <0.05. All data analyses were conducted using a statistical software program, specifically SPSS.
This study involved human subjects, with fetuses as the primary focus. Ethical considerations were addressed in accordance with the guidelines of the Declaration of Helsinki, and approval was obtained from the ethical committee (Approval number: 333/KEPK/2021). All medical information related to the research is kept confidential. Participants had the right to decline involvement if they chose not to participate. The researcher covered all expenses associated with the study, including any additional costs incurred.
3. RESULTS
A total of 400 pregnant women in their first trimester (4-13 weeks) underwent ultrasound examinations for check-ups. The distribution of characteristics among the study participants is presented in Table 1.
Table 1 illustrates that the average age of the participants fell within the reproductive range, specifically between 21 and 30 years old. A significant portion of the subjects were experiencing their first pregnancy, classified as primigravida, accounting for 33%. The majority of the mothers had completed high school education, representing 57.75%, and were actively employed.
| Characteristic | X±SD |
|---|---|
| Age (year) | 22.49 ± 4.5 |
| <20 | 11 |
| 21-30 | 301 |
| ≥30 | 88 |
| Parity | - |
| 0 | 232 (33) |
| 1 | 92 (23) |
| 2 or more | 76 (19) |
| Education | - |
| No college | 1 (0.25) |
| JHS* | 42 10,5) |
| SHS* | 231 (57,75) |
| College Graduate | 126 (31,5) |
| Job Status | - |
| Civil servant | 103 (25.75) |
| Private employee | 147 (36,75) |
| Entrepreneur | 95 (23.75) |
| Unemployed | 55 (13,75) |
Fetal growth measurements from the LMP compared to first-trimester ultrasound assessments in the Minangkabau ethnic group are detailed in Table 2.
| Weeks | GS Diameter (Mean ± SD) |
|---|---|
| 4 | 0,87 ± 0,36 |
| 5 | 1,06 ± 0,38 |
| 6 | 1,81 ± 0,37 |
| 7 | 2,47 ± 0,53 |
| 8 | 3,46 ± 0,84 |
| 9 | 4,09 ± 0,47 |
| 10 | 4,52 ± 0,37 |
| 11 | 5,09 ± 0,90 |
| 12 | 6,04 ± 0,41 |
Table 2 presents the average GS diameter based on the LMP. It was observed that the GS diameter increased over time, with an average measurement of 1.06 ± 0.38 cm at 5 weeks and 4.52 ± 0.37 cm at 10 weeks.
According to Fig. (1), there was a significant increase between gestational age and LMP.
Table 3 illustrates the average biometric measurements of gestational age (GA) based on the LMP. It was noted that there was an increase in the mean length of the CRL, with the average measurements being 1.33 ± 0.34 cm at 7 weeks and 3.66 ± 0.37 cm at 10 weeks.
Based on Fig. (2), there was a significant increase between gestational age and LMP.
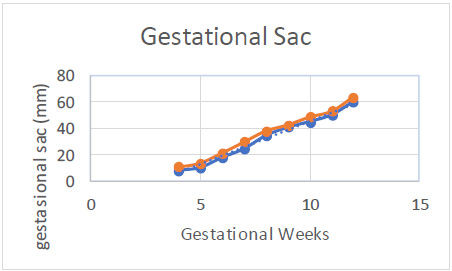
Mean measurements of the parameters GS according to LMP.
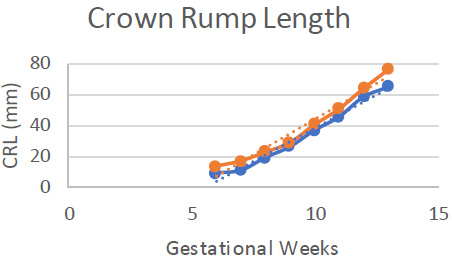
Median measurements of the parameters CRL according to LMP.
| Weeks | CRL (Mean ± SD) |
|---|---|
| 6 | 0,86 ± 0,48 |
| 7 | 1,33 ± 0,34 |
| 8 | 1,87 ± 0,38 |
| 9 | 2,58 ± 0,29 |
| 10 | 3,66 ± 0,37 |
| 11 | 4,54 ± 0,51 |
| 12 | 5,85 ± 0,53 |
| 13 | 6,50 ± 1,10 |
The results of fetal biometric measurements obtained via ultrasound for each participant during the first trimester of pregnancy indicated an increasing variability with advancing gestational age for both measured variables.
The relationship between fetal growth based on the LMP and first-trimester ultrasound assessments within the Minangkabau ethnic group is presented in Table 4 below.
| Parameter | Mean ± SD | r | p-Value | R2 |
|---|---|---|---|---|
| GS | 4,59 ± 1,30 | 0,89 | 0,005 | 0,90 |
| CRL | 3,02 ± 1,33 | 0,94 | 0,0001 |
According to Table 4, a robust correlation (r=0.94) was observed between the CRL diameter and GA based on the (LMP, with a p-value of 0.0001, indicating statistical significance.
4. DISCUSSION
We conducted a study on Minangkabau women, emphasizing fetal growth at both the population and individual levels. This prescriptive approach aimed to develop appropriate growth standards for the gestational sac (GS) and crown-rump length (CRL), as well as to estimate gestational age during the first trimester of pregnancy.
A related study examining first trimester growth in relation to ethnicity was previously conducted by Bottomley C et al., focusing on women of black and white ethnic backgrounds. The research indicated that crown-rump length (CRL) is utilized for dating pregnancies, which subsequently affects growth assessments. The authors suggested that it is important to consider individualized growth charts that account for maternal factors impacting growth during the first trimester [25].
Our research possesses several significant methodological and conceptual advantages. Firstly, it specifically targets the Minangkabau ethnic group within Indonesia's diverse multiethnic population, including children from mixed parentage, which adds relevance to the findings. Secondly, a standardized protocol was implemented for participant recruitment and data gathering. Thirdly, the study was population-based and focused solely on naturally conceived singleton pregnancies with confirmed LMPs and classified as low-risk. Lastly, all participants were monitored throughout late pregnancy, with exclusions made for cases of fetal or neonatal death and severe pregnancy complications [11-14].
This study included 400 pregnant women in their first trimester. The demographic profile of the participants revealed that the average age of the women ranged from 21 to 30 years, with 33% experiencing their first pregnancy. Additionally, 57.75% of the participants had completed senior high school, and many of them were employed. These characteristics are important determinants of fetal growth.
The gestational sac is the earliest structure typically visualized through imaging in the first trimester. Transabdominal ultrasound can detect this structure as early as 5 weeks of gestational age, while transvaginal ultrasound may reveal it as soon as 4 weeks. Our findings indicate a growth in the average diameter of the gestational sac, with the mean diameters recorded at 5 weeks and 10 weeks being 10.6 ± 3.8 mm and 45.2 ± 3.7 mm, respectively. Experts suggest that the average length of the gestational sac at 5 weeks is approximately 10-16 mm, while the diameter at 10 weeks is between 45-49 mm [3, 11] This data suggests a slight variation in measurements between Indonesian and American populations [2, 18].
We observed an increase in the average CRL, with the mean measurements recorded at 7 weeks and 10 weeks being 1.03 ± 0.34 cm and 3.66 ± 0.37 cm, respectively. According to MacGroger et al., the mean CRL length at 7 weeks ranges from 1.0 to 1.1 cm, while at 12 weeks, it typically measures between 5.1 and 5.9 cm [10]. Similarly, Robinson and Fleming et al. reported that the mean CRL length at 7 weeks is between 1.0 and 1.5 cm and at 12 weeks is around 5.6 to 5.9 cm [26]. Comparing our findings with previous studies suggests a slight size variation between the Minangkabau ethnic group in Indonesia and American populations. A limitation of our study is its focus on a single ethnic group, which may affect the establishment of a more accurate national standard. Given Indonesia's diverse ethnic composition, we recommend further research that includes other ethnic groups to enhance the accuracy of national growth standards.
According to the data presented in Table 4, there exists a significant correlation (r=0.94) between the CRL diameter and GA based on the LMP, with a p-value of 0.0001. Although there are potential sources of error, CRL measurement remains a reliable and effective method for determining gestational age during the first trimester. This finding is supported by various studies indicating that CRL is the most accurate technique for estimating gestational age [1, 3, 27].
The findings from our current study on fetal biometry in the first trimester complement the insights gained from our previous research involving 520 pregnant women in their third trimester within the Minangkabau ethnic group. In the earlier study, we observed distinct fetal growth characteristics that highlighted the importance of monitoring growth patterns throughout pregnancy [28, 29]. The strong correlation established between CRL and gestational age in the first trimester serves as a foundational benchmark for early fetal development, which is crucial for predicting later growth trajectories. By comparing these early measurements with the growth parameters identified in the third trimester, we can better understand the continuity of fetal development and the potential implications for prenatal care. This integrated approach underscores the necessity of establishing comprehensive growth standards that span all trimesters, allowing healthcare providers to identify deviations from expected growth patterns early on and implement timely interventions. Furthermore, as the Minangkabau ethnic group exhibits unique growth characteristics, our findings advocate for the development of tailored growth charts that reflect the specific needs of this population, ultimately enhancing maternal and fetal health outcomes.
A limitation of our study is its focus on a single ethnic group, which, while useful for establishing tailored growth standards, limits generalizability across Indonesia's diverse populations. Genetic factors and environmental influences unique to the Minangkabau, such as dietary habits and regional climate, may also affect fetal growth patterns in ways that may not apply to other ethnic groups. Additionally, socioeconomic factors specific to this population, including healthcare access and maternal education levels, might influence fetal growth outcomes. Acknowledging these factors is important, as they may limit the applicability of our findings to Indonesia’s broader population.
Future research should include longitudinal studies that track fetal growth throughout all trimesters and into infancy, which could reveal how early differences in fetal biometry impact later development. Expanding studies to include other major Indonesian ethnic groups, such as Javanese or Balinese, would also enable a more comprehensive national standard that considers Indonesia's ethnic diversity. Such studies should ideally examine the interplay of genetics, environment, and socioeconomic factors across these populations to establish more nuanced and widely applicable growth standards.
CONCLUSION
The key outcome of this research indicates that crown-rump length (CRL) serves as an accurate and effective method for evaluating gestational age during the first trimester. We have established a national prescriptive standard for the initial assessment of fetal growth and first-trimester ultrasound dating of pregnancy, specifically tailored for use in Indonesia, with a focus on the Minangkabau ethnic group. This is a single ethnic group study, and Indonesia has a variety of ethnicities, and we recommend to study other ethnic groups in Indonesia to obtain a more accurate national standard.
These findings have significant implications for clinical guidelines, as they provide healthcare professionals with population-specific benchmarks that improve the accuracy of pregnancy dating and early fetal growth assessment. This can help to reduce the risk of misclassification of fetal development stages and support more precise monitoring of pregnancies, ultimately contributing to better maternal and neonatal outcomes. In terms of healthcare training, the study underscores the need for ultrasound training programs that incorporate these population-specific standards, enabling practitioners to apply these tailored metrics in practice effectively. Additionally, resource allocation in healthcare could benefit from adopting these findings, as this new standard may guide the prioritization of ultrasound resources and services, particularly in areas with high Minangkabau populations, ensuring that accurate prenatal care is accessible to more communities.
AUTHORS’ CONTRIBUTION
Y.: Performed material preparation and data collection; Y., J.S., B.I.U., and P.S.: Contributed to the study conception, design and analysis; Y.: Wrote the first draft of the manuscript. All authors read and approved the final manuscript.
LIST OF ABBREVIATIONS
| USG | = Ultrasonography |
| CRL | = Crown-Rump Length |
| GA | = Gestational Age |
| NICHD | = National Institute of Child Health and Human Development |
| AC | = Abdominal Circumference |
| SGA | = Small for Gestational Age |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study has been approved by Health Research Ethics Committee RSUP Dr. M. Djamil Padang, Indonesia (approval number: 333/KEPK/2021).
HUMAN AND ANIMAL RIGHTS
All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
ACKNOWLEDGEMENTS
A preprint has previously been published. We want to thank all staff at Hospital M. Djamil Padang. They facilitated us in data collection and all pregnant women participants who had been willing to participate in this research.


