All published articles of this journal are available on ScienceDirect.
DDH Diagnosis: What do we Know so Far?
Abstract
Background:
Developmental Dysplasia of the Hip (DDH) is a common disease worldwide. The success of treatment depends on making an early and accurate diagnosis using clinical examination and imaging. Diagnosis of DDH patients is challenging and controversial, and many cases still present late. There is still a lack of consensus among health care providers regarding the diagnostic methods in terms of interpretation, accuracy and appropriate timing. The clinical practice seems to widely vary between healthcare providers in different parts of the world.
Aims:
We aim to provide a clear pathway to help healthcare professionals from different disciplines in diagnosing DDH at the earliest age possible in order to start an effective treatment and avoid complex procedures and unnecessary complications. We also aim to provide an extensive review that will provide a standardized reference detailing the whole diagnostic process in order to improve the clinical outcome and save resources.
Methods:
We reviewed the relevant literature using Pubmed, Google Scholar, and the Cochrane library and looked into the references lists of all of these papers to gather all the available evidence. All papers were carefully critiqued to use the most rigorous in the conclusions of this review. We included articles discussing clinical examination and radiological diagnosis. Papers discussing the most used clinical examination tests and the timing of clinical evaluation were reviewed. Also, we looked into papers discussing radiological diagnosis using ultrasound scanning and standard radiographs in terms of sensitivity, specificity and reliability and their relation to patient’s age and other variables.
Results:
DDH diagnosis must start at a very young age by repeated examination using specific clinical tests looking mainly for signs of major instability. Ultrasound scanning is the investigation of choice in screening and early diagnosis, and the Graf method seems to be the most reliable method. X-rays are not reliable in early diagnosis, especially in the first four months of life, and can lead to over-diagnosis, particularly when using the Acetabular Index as the sole measurement tool.
Conclusion:
DDH diagnosis has many controversies, and the practice is still widely variable. This review provides an evidence-based, updated pathway for early DDH diagnosis in order to improve the clinical outcome, avoid complications, reduce the need for surgical treatment and save public resources. It highlights some malpractices that are still used in the diagnostic process. The review provides a standardized reference to doctors from different specialties and with different experience levels who are tasked with assessing children for DDH.
1. INTRODUCTION
DDH is a common disease with a wide spectrum of severity that can range from mild under-development of the acetabulum ‘Dysplasia’ to full dislocation of the joint [1]. From population-based studies, it was concluded that around 75-85% of newborns have morphologically normal hips, 13-25% have immature, while 2-4% have dysplastic hips [2]. DDH incidence varies due to geographical, genetic and cultural factors but generally ranges from 0.006 in Africans to 7.6% in Native Americans [3]. Most cases have dysplasia, and only 10% of patients have a complete dislocation, i.e., around 1:1000 [4]. DDH is a painless and silent disease in infants and young children and therefore can easily be missed by the parents [1, 5].
DDH must be checked for in all newborns but particularly in those with risk factors including breech position, those with a positive family history in a first-degree relative, female babies, primiparity ‘first baby to the mother’, oligohydramnios, post-term babies, birth weight more than 4 Kgs, babies with other packaging disorders indicating a mismatch between the size of the baby and the intrauterine volume like torticollis and metatarsus adductus, etc. The only postnatal risk factor is tight swaddling, where the baby is wrapped with the hips and knees fully extended and adducted, which is a common cultural practice in many parts of the world.
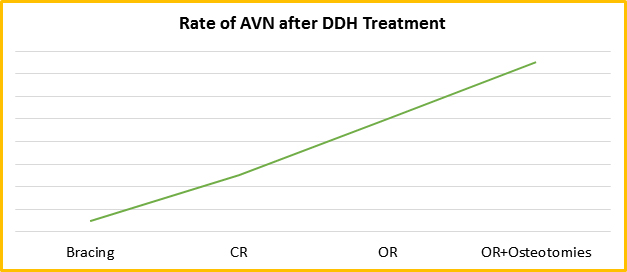
The treatment of DDH is usually guided by the age of the patient. Generally, infants under 6 months of age are treated conservatively with braces, e.g., Pavlik harness. Children who present between six and twelve months of age (and up to Eighteen Months in some papers) and those who fail the bracing treatment require a closed hip reduction under general anaesthesia guided by an arthrogram and using a spica cast for immobilization providing the hip is reducible and stable. Older children and those who fail the above treatment options require surgical open reduction with a spica cast, and with advancing age, pelvic and/or femoral osteotomies may be added as well to achieve congruent reduction and stability. These treatment options, however, are not completely risk-free and can carry the risks of nerve injury and Avascular Necrosis (AVN) of the femoral head. Even the normal ‘untreated hip’ hip can be affected by AVN at times due to mal-positioning of the hips in the brace or spica cast. The rate of AVN increases with the increasing complexity of the treatment (Fig. 1). Also, spica casts can cause intestinal obstruction ‘Spica syndrome’ if not appropriately applied.
The earlier a growing hip is put into its normal place, the higher the chance of allowing normal growth of the joint and avoiding unwanted consequences [6, 7]. At a very young age, the treatment is usually simpler and more successful, with less complications and less conversion rates to surgery [8-10]. Early detection and treatment provide the best chances for successful conservative treatment [11] and the lowest risk (1%) of developing iatrogenic avascular necrosis [12]; a dreaded and known complication to DDH treatment that can cause a long-lasting disability. Children treated late for DDH are at higher risk of developing complications [13]. The rates of surgical open reduction are much lower if the diagnosis and treatment are initiated by 6 weeks of age compared with 4 months of age, for example [14]. First presentation beyond 3 months of age is considered late by some authors [15, 16].
Untreated DDH leads to a significant increase in the rates of osteoarthritis in adulthood with a significantly earlier age of onset [10, 17]. Total hip arthroplasty is a very common procedure in adults with under-treated DDH [18]. According to the Norwegian arthroplasty registry, one-third of THA operations under the age of 65 were performed for osteoarthritis secondary to under-treated DDH [18]. On the other hand, overdiagnosis and over-treatment can lead to significant complications like avascular necrosis [19, 20] and femoral nerve palsy [21].
DDH Diagnosis can be made by one or more of the following: Clinical examination, ultrasound scanning and X-Rays radiographs. In this review, we will discuss the available literature discussing the diagnostic methods in detail to find the most up-to-date diagnostic strategy for early and correct diagnosis in order to start early treatment to optimize the outcome and avoid future complications or complex procedures.
There are many papers in the literature discussing different aspects of the diagnostic process of DDH. However, these papers often discuss specific points in the diagnostic process and seldom give a comprehensive and detailed overview of the whole process. Most of the evidence is designed by and directed at orthopaedic surgeons and/or paediatricians. However, in many parts of the world, this task is for physicians from other disciplines to perform, e.g., family health practitioners, public health doctors, and sometimes newly qualified doctors with little clinical experience.
In the developing world, public healthcare systems are overwhelmed and suffer from a lack of resources and sub-specialization, and these cases very often get missed and present late. DDH screening programs are being introduced in some countries, but often, the doctors providing the service do not have an orthopaedic background and sometimes do not have the required knowledge and clinical skills. The lack of expertise in ultrasound scanning forces the healthcare provider to order standard X-ray radiographs for diagnosis -which is thought to be easier to interpret which carries the risk of diagnostic errors. This leads to recurrent unnecessary visits and repeated X-rays to confirm the diagnosis with increased radiation exposure. This causes over-diagnosis and over-treatment, which has negative clinical and psychological implications on the child and the family or under-diagnosis, leading to increased rates of surgical intervention and long-term disability. All of this drains the already scarce public health resources and increases the workload on already exhausted health systems.
This review provides a comprehensive and detailed guide to healthcare providers in all disciplines involved in DDH diagnosis. It provides the necessary knowledge for physicians at all levels regarding the disease and its diagnosis and treatment options. It outlines the necessary clinical tests, their technique and the age at which the test is performed. It also provides a pathway for diagnosis using examination and radiological imaging and provides the necessary knowledge regarding ultrasound scanning and X-rays with their advantages and disadvantages.
2. METHODS
We reviewed the medical literature via searches on PubMed, Google Scholar and the Cochrane library. We used all search words related to DDH diagnosis, screening programs, clinical examination, specific examination tests, ultrasound and X-ray assessment of paediatric hips, etc. Also, we looked at papers discussing national screening programs in many countries. We also reviewed all of the references lists of these papers and used all the relevant ones in this review. 97 papers were found with relevant information on the above topics after ignoring the ones with poor methodology. All papers were read and critiqued thoroughly and relevant information extracted. The gathered information was used to draw an evidence-based review of the whole process of DDH diagnosis, which can be a guide to healthcare providers from all disciplines who can be tasked with assessing a child for DDH.
3. RESULTS AND DISCUSSION
3.1. Clinical Examination
Clinical examination is a very important part of the diagnosis [1]. However, because DDH has a wide spectrum, not all DDH cases can be detected clinically, particularly in patients with dysplasia only without instability [22]. This is often overlooked, and many doctors rely on imaging alone for diagnosis or rely on clinical findings of other physicians without examining the child themselves. The age at which the baby is examined is very important. Many babies will have ‘immature’ hips at birth; i.e., dysplastic acetabulae and/or mild instability, which is physiological for age, and most of these hips will eventually ‘mature’ and become normal over a period of few weeks on both clinical examination and ultrasound [23, 24]. High rates of resolution without intervention (90%-97%) have been reported in this age group [23, 25]. The hips that fail to mature over time need treatment to avoid future complications.
Examining a baby for DDH is extremely difficult and requires a great deal of training and experience [24, 26, 27]. It was found that the ability of the examiner to find positive signs is better in the outpatient setting compared with the neonatal ward immediately after birth [28, 29]. Clinical examination performed by orthopaedic surgeons possibly detects more DDH cases compared to paediatricians, but the difference is not statistically significant [13]. There is a fair evidence to include serial clinical examination of the hips by a trained clinician in the periodic health examination of all infants until they are walking independently [30, 31]. This will identify patients who were initially missed by previous screening [7, 31-34].
There is good evidence to support a period of close observation for newborns with clinically detected DDH, as mentioned above. A positive Barlow test at birth may resolve spontaneously over the first few weeks of life [35]. However, there is insufficient evidence to determine the optimal duration of observation [30]. It is thought that 71% of hips stabilize within 2 weeks and 88% within 1 month after birth [36, 37]. Delaying the treatment by 30 days in borderline cases was shown not to affect the outcome of treatment [24, 38]. It is suggested that examination at 6 weeks of age is more specific in detecting true instability [39].
Limited hip abduction, in addition to positive Barlow and Ortolani tests, is an important sign in DDH [40]. A ‘clunk’ rather than a ‘click’ is important while performing the ‘Ortolani’ and ‘Barlow’ tests [1, 3, 39, 41]. This is often confusing and the distinction greatly depends on the experience of the examining physician. The best way to describe the difference is that a clunk must be palpated rather than heard, indicating dislocation or re-location of the joint. An audible hip ‘click’ on its own is not considered a sign suggestive of gross hip instability [10, 41]. The Barlow and Ortolani tests must be done very gently [42, 43]. Both tests are performed in the supine position with both hips and knees flexed at 90 degrees. In the Barlow test, the stability of the located hip is checked. The examiner tries to dislocate the hip from the reduced position by applying a gentle posterior axial force in slight hip adduction while feeling for a clunk. The Ortolani test assesses the reducibility of an already dislocated hip; the hip is abducted with a gentle anterior force applied to the greater trochanter. Again, a clunk suggests that the hip was dislocated but is reducible [3]. The sensitivity of the Barlow test in experienced hands is 87-97% [30]. Barlow and Ortolani tests become less reliable with the advancing age of the baby [3, 30, 42, 44]. It is important to note that negative findings on the Barlow and Ortolani tests do not rule out DDH.
Asymmetrical hip abduction becomes the most reliable sign to diagnose dislocated hips with the increasing age of the baby [3, 20, 44, 45]. Abduction less than 75 degrees can be suggestive of DDH [3]. 30% of infants with limited hip abduction will have confirmed DDH on USS [28]. In particular, unilateral limitation of abduction (asymmetrical abduction) has a very strong positive predictive value for DDH [28]. Furthermore, limited abduction seems to be the best clinical sign to be used for screening purposes by non-orthopaedic surgeons like paediatricians and family medicine specialists, etc. [45]. This sign is important in all age groups. However, in cases of bilateral dislocations, the abduction will appear symmetrical, which can be deceiving for the examiner. In this case, the range of abduction (Normal > 75 degrees) can be used to make the judgment.
Leg length discrepancy (LLD) can be suggestive of dislocation in one of the hips, although there are many other causes. This is checked by performing the Galleazzi-Allis test. The hips and knees are bent, and the feet are placed next to each other. Looking from the side, the tibial tuberosities must be at the same level. If not, this indicates that one femur is shorter than the other or that the ipsi-lateral hip is dislocated [3]. In the case of bilateral hip dislocations, this test may not pick any leg length difference.
Some less significant signs that may indicate DDH are asymmetrical thigh or groin creases and ‘clicky hips’. These signs have a lower predictive value for DDH compared to the signs described above. For example, asymmetrical thigh creases or gluteal folds can be present in 27% of babies with normal hips [3].
After walking age, the gait can become affected if the child has a dislocated hip. Trendelenberg gait is caused by abductor muscle insufficiency secondary to DDH or to other causes occasionally. In cases of bilateral hip dislocations, the ‘waddling gait’ will result, which is basically a bilateral Trendelenberg gait. LLD can cause toe walking on the affected side due to shortening ‘unilateral toe walking’ or a ‘flexed knee’ gait on the normal side. As a result of long-standing hip dislocation, a fixed flexion contracture of the hip may develop, leading to increased lumbar lordosis in older ages [3]. The clinical examination tests for DDH are summarized in Table 1.
3.2. Ultrasound Scanning (USS)
USS is the screening method of choice in terms of reliability and cost to diagnose DDH [46, 47]. USS is superior to X-Rays in the first six months of life due to the late ossification of the proximal femur in infants [20]. USS is known to detect more cases of DDH compared to clinical examination alone [23, 28]. In fact, paediatric orthopaedic surgeons seem to think that clinical examination alone is suboptimal for assessing children for the possibility of DDH and that USS assessment is essential [48]. Combining clinical examination with USS is advisable for DDH [29]. The reason for this is that USS will detect acetabular dysplasia (which constitutes most of DDH cases), which is usually missed by clinical examination due to the lack of instability [49, 50]. Performing a USS on children with clinically detected instability is also recommended, as this will reduce the number of children treated with braces unnecessarily without increasing the overall cost [51]. It also provides a baseline image for comparison throughout treatment in positive cases.
Most cases of neonatal hip dysplasia identified by USS will resolve in the first few weeks of life, as discussed above [20). There is little consensus among orthopaedic surgeons regarding what constitutes a normal or abnormal USS in neonates [24, 52, 53]. Performing the USS under six weeks of age may give a false positive result leading to overdiagnosis and over-treatment [44]. It is agreed that in the absence of clinical instability, the first USS must be done at the age of 6 weeks in full-term babies. In preterm babies, it is recommended to use the corrected age for ultrasound screening, and the corrected age should be preferably > 44 weeks of gestation at the time of the first scan in the absence of clinical instability, e.g., if the baby is born prematurely at 34 weeks of gestation, the first USS must be done at 10 weeks of age instead of six weeks (44-34=10) [54]. The use of USS reduces treatment rates for ‘over-diagnosed’ DDH and its associated physical and psychological complications on the baby and the parents [21].
| - | Principle of the Test | Technique | Upper Age the Test Can Be Used | Remarks |
|---|---|---|---|---|
| Barlow | Assess stability of the located hip | Hips & knees flexed 90 degrees. Posterior axial force to hip. Palpable clunk on dislocation | 3-4 months | -Requires lots of training and experience to perform. -Subtle instability can be physiological in the first 6 weeks of life. -Negative test does not rule DDH out. |
| Ortolani | Assess reducibility of the dislocated hip | Hips & knees flexed 90 degrees. Abduct hip with anterior force to greater trochanter. Palpable clunk on relocation. | 3-4 months | -Requires lots of training and experience to perform. -Negative test does not rule DDH out. |
| Galleazzi Allis | Assess LLD, which can be caused by unilateral DDH | Flex hip 45 degrees and knee 90 degrees. Check heights of tibial tuberosities from the side to check for limb shortening. | All ages | -This test is not specific to DDH and be positive in other pathologies. -May be negative in bilateral DDH |
| Reduced Abduction | Assess the restriction in abduction commonly affected by DDH | Abduct hips in flexion. Hips usually abduct > 75 degrees. Watch for symmetry. | All ages | This test can be deceiving in bilateral dislocations |
| Trendelenberg Gait | Assess for abductor insufficiency during the stance phase of the gait cycle | Watch gait. The pelvis dips down while standing on the affected limb and the trunk sways towards the other side. | After walking age | This gait is not specific to DDH and can be caused by other pathologies |
| Waddling Gait (Bilateral Trendelenberg Gait) |
Assess for abductor insufficiency during the stance phase of the gait cycle | Watch gait. The pelvis dips down while standing on the affected limb and the trunk sways towards the other side. Finding present on both sides. |
After walking age | This gait is not specific to DDH and can be caused by other pathologies |
| Unilateral toe walking gait | Assess LLD, which can be caused by DDH | The child walks on his toes without a heel strike on the affected side which has apparent shortening | After walking age | -In cases of bilateral dislocations this sign is absent. -This gait is not specific to DDH and can be caused by other pathologies |
| Flexed knee gait | Assess LLD, which can be caused by DDH | The unaffected knee remains flexed while standing due to the relatively longer limb | After walking age | -In cases of bilateral dislocations this sign is absent. -This gait is not specific to DDH and can be caused by other pathologies |
| Fixed flexion hip deformity and increased lumbar lordosis | Positive Thomas Test indicating chronic dislocation. | The affected hip lacks full extension with flexion of the contralateral hip. | Older children with neglected DDH | This gait is not specific to DDH and can be caused by other pathologies |
| Type | Description | Bony Roof | Bony Rim | Cartilage Roof | Alpha Angle | Beta Angle |
|---|---|---|---|---|---|---|
| I | Mature Hip | Good | Angular/Blunt | Covers the Femoral Head | ≥ 60 | <77 |
| IIa | Physiological (<3months) | Deficient | Rounded | Covers the Femoral Head | 50-59 | >55 |
| IIb | Delay of Ossification (>3months) | Deficient | Rounded | Covers the Femoral Head | 50-59 | <55 |
| IIc | Critical Hip | Severely Deficient | Rounded to flattened | Still Covers the Femoral Head | 43-49 | <77 |
| D | Decentering Hip | Severely Deficient | Rounded to flattened | Displaced | 43-49 | >77 |
| III | Dislocated Hip | Poor | Flattened | Pressed Upward, perichondrium slopes cranially | < 43 | >77 |
| IV | Dislocated Hip | Poor | Flattened | Pressed downward, perichondrium is horizontal or dips caudally | <43 | - |
There are many USS screening methods for DDH, e.g., Graf, Harcke, Suzuki methods, etc. [55-59]. There is some fair agreement in the literature that the Graf method (Table 2) is the most reliable for scanning [47, 48, 60]. However, some studies showed that patients with ‘borderline normal’ alpha angles according to the Graf method may need some attention to other signs like femoral head coverage (Harcke method) or follow up with X-Rays to make sure hips do not need treatment [61]. Despite that, the correlation between alpha angles and femoral head coverage is strong, meaning that most children with normal alpha angles, according to Graf, will also have a very good femoral head coverage (>50%) according to Harcke [62].
Standardization of teaching and training of USS examination in general [63] and of the Graf technique, in particular, is mandatory [48]. The Graf method of scanning and classifications needs to be taught by a structured programme to increase inter-rater reliability [64]. Repeatability must be achieved between examiners at the time of recording the appropriate images (performing the scan) and at the time of reading (interpretation of images) [2]. This is of paramount importance in any public health service where patients can often be seen by different physicians during follow-up visits.
The specificity and accuracy of USS are lowest before 28 days of age due to the ‘Normal immaturity of the hip’ and highest at three months of age [25, 65, 66]. Therefore, a two-step US screening of newborns is recommended: after the end of the first month (around six weeks of age) and within the fourth month of life [67]. For mild dysplasia without instability, doing a delayed USS at 6 weeks of age reduces the rates of overtreatment without increasing the numbers of late presenting cases [24]. Babies with a normal clinical examination and normal ultrasound at 6-8 weeks are considered normal and do not require further radiographic imaging for evaluation of DDH [66]. The false-negative rate of USS is nearly zero %, i.e., it is unlikely to miss cases of DDH [15]. If the USS shows a Graf Type II c or above, treatment is started, but if the USS shows a type IIa hip, then it must be repeated at 3 months of age ‘The second step’ [55, 56, 68]. Early USS diagnosis is associated with significantly reduced rates of open reduction for DDH due to rapid initiation of brace treatment [14].
The upper age for performing USS reliably is still controversial. Graf recommended using USS up to the age when the ossific nucleus develops enough to obscure the visualization of the lower limb of the acetabulum. This age differs between different babies, but many studies stated that USS can be reliably used up to 6-8 months of age [5, 28, 69-73]. USS can be used at 6 months of age with a very high negative predictive value comparable to X-Rays [66]. Successful conservative management and avoidance of surgery depend on reaching an early diagnosis, preferably in the first three months of life, which can only be reliably achieved using USS.
3.3. X-ray
X-ray is used for late diagnosis of DDH, and its use in screening for DDH is now historical [24]. Pelvis antero-posterior X-ray becomes helpful after the 4th month of life [42, 74-76] and is the investigation of choice to diagnose DDH after 6-9 months of age [5, 77]. The reliability of X-ray increases with the increasing age of the baby [78, 79]. It is suggested that using X-rays in children younger than 7 months of age is unreliable [78].
Getting an optimal position of the hips while getting the X-ray is of paramount importance for making correct measurements and diagnoses. In one study of 76 children (between 8 and 11 months of age) who had pelvic radiographs, only 54% of radiographs were taken optimally [1]. Suboptimal radiographs will inevitably lead to a wrong diagnosis and wrong treatment and must be repeated to avoid making a wrong diagnosis [1].
There is still a lack of consensus on the definition of clinically relevant DDH on radiographs [30, 80]. Radiographic signs of DDH include increased acetabular index [33], disruption of Shenton’s line, widened pelvic floor, delayed appearance of the femoral ossific nucleus, lateral migration of the femoral head and decreased femoral head coverage [80-82]. The acetabular index (AI), in particular, is widely used to assess patients with dysplasia. This is especially true in countries and areas that do not have access to ultrasound scans due to a lack of resources and/or expertise. Also, physicians diagnosing these cases often do not have a paediatric or orthopaedic background and therefore, the easiest way of XR assessment to them is to measure the AI. However, there is a poor inter-observer and intraobserver reliability of the AI and it can be easily affected by pelvic tilt and rotation and by the presence or absence of an acetabular notch [78, 79, 83-85]. The difference in AI measurement can be altered by 6-12 degrees if the tilt or rotation is not correct [86]. This can change the diagnosis and the treatment decision. If there is an abnormal tilt or rotation, this can over- or under-estimate the AI [87]. The normal AI values are not constant to all and depend on age, sex and the side-left or right [83]. A single radiographic measurement may not be accurate, but the change of value over series of images is more reliable [84], which can obviously delay the diagnosis. This unreliability is caused by the fact that the acetabulum has a big cartilaginous part in young children that is not visible on X-ray therefore can mislead the interpreter [88, 89]. Also, the lateral aspect of the acetabulum is irregular in dysplastic hips, which makes the AI measurement more difficult [80, 90]. In fact, there is a debate whether X-Ray variability, e.g., increased AI constitutes an actual disease or not [91].
For subluxed and dislocated hips, there are many classifications to determine the severity of the disease, like the Tonnis classification [92, 93], the International hip Dysplasia Institute (IHDI) classification [5, 72] and others [94, 95]. The IHDI classification seems to have the best inter and intra observer reliability and can be used without the presence of the ossific nucleus [72].
At young ages, X-Rays have poor inter-observer and intra-observer reliability [30, 84]. The more dysplastic the hip is, the less the inter-rater reliability [86]. Nonetheless, radiation exposure needs to be taken into consideration, especially with repeated radiographs which are often needed for diagnosis and treatment follow-up. The radiation exposure associated with X-rays must be kept to a minimum [96]. The correlation between Graf’s alpha angles on USS with the Acetabular Index of Tonnis is poor [62]. So is the correlation between Harcke’s femoral head coverage with AI, especially in borderline cases [88]. This confirms that relying on AI is not going to give an accurate diagnosis compared to the gold standard USS and questions the reliability of the X-rays altogether, having learned that many doctors use AI as the primary radiographic diagnostic measurement.
Despite the above, X-rays may be an option for screening and diagnosis of DDH in areas without easy access to USS [97]. The age group between 4-6 months is controversial, and both USS and X-Rays can be used for DDH diagnosis [1]. At this age, X-Rays have a good negative predictive value to rule out DDH but a very poor positive predictive value; if X-rays are normal at this age, DDH is ruled out, however, if the X-Ray is suggestive of DDH, a USS must be obtained to confirm the diagnosis before starting treatment during follow up visits.
On many occasions, especially under the age of 18 months, X-Rays do not provide the necessary details to decide on the next step in management. This is mainly due to delayed ossification of the hip joint. This can be overcome by performing a hip arthrogram to outline the anatomy and the stability. The arthrogram is done under general anaesthesia. A small amount of contrast material is injected inside the hip capsule, and the joint is screened under a c-arm image intensifier to outline the anatomical structures and the stability of the joint.
CONCLUSION
DDH diagnosis is not easy and has many controversies. Clinical examination shortly after birth is aimed at detecting signs of major instability. Barlow, Ortolani, Galleazzi Allis tests and reduced abduction are the signs to look for at this age. If any of these signs are present, USS must be obtained soon and treatment is started if DDH is confirmed. Late clinical signs of DDH include gait abnormalities, e.g., Trendelenberg, Waddling, unilateral toe walking and flexed knee gaits in addition to positive Thomas test with secondary increased lumbar lordosis in chronic cases.
If the examination does not show any instability, but there are other signs, e.g., asymmetrical thigh creases or ‘clicky hips’, or one or more risk factors for DDH are present, then a USS must be obtained at 6 weeks of age to avoid the risk of over-diagnosis associated with the normal laxity and dysplasia in the first few weeks of life. A normal USS (Type I hips) at any age rules DDH out. In Type IIA hips ‘immature hips’ the scan must be repeated at 3 months of age. Type IIb hips and above are abnormal and require immediate treatment. USS remains the gold standard for diagnosis until 6 months of age.
X-rays become reliable after 6 months of age, but from the age of 4 months and above, a normal Hip X-ray can exclude DDH. A child with an abnormal X-Ray between 4-6 months of age needs to be further assessed by a USS to confirm the diagnosis. The Acetabular Index of Tonnis is a very commonly used radiographic measurement, but the literature suggests that its reliability is questionable with poor correlation with the USS measurements.
At older ages, children with dislocated hips will still have reduced hip abduction and leg length discrepancy as well as an abnormal gait as above. X-Rays are the investigation of choice after the age of 6-9 months. However, at times even x-rays cannot provide the necessary details to decide on the next step in management. This is mainly due to delayed ossification of the hip joint. This can be overcome by performing a hip arthrogram to outline the anatomy and the stability.
This review provides a complete reference guide to doctors from different disciplines and with different experience levels involved in DDH diagnosis. It highlights the importance of learning the principles and the proper techniques of clinical assessment and radiological imaging. Following a clear pathway for DDH diagnosis standardizes the practice in public health systems and clearly gives a better clinical outcome and saves resources. This review is based on the available literature, which still has controversies in some areas. Future research is needed to clear some of the ambiguities, but for now, we believe that this review constitutes an up-to-date useful reference for DDH diagnosis, and we recommend its use in clinical practice.
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


