All published articles of this journal are available on ScienceDirect.
Migraine and Multiple Sclerosis Across the Female Reproductive Lifespan: Insights for the Obstetrician-Gynecologist and Neurologists: A narrative review
Abstract
Introduction
Migraine and Multiple Sclerosis (MS) are chronic neurological conditions that predominantly affect women, especially during their reproductive years. These disorders are influenced by hormonal fluctuations, particularly estrogen, which plays a crucial role in immune modulation, neuronal excitability, and vascular function.
Objective
This review examines the relationship between migraine, MS, and female reproductive health, with a focus on hormonal influences throughout critical life stages: puberty, menstruation, pregnancy, postpartum, and menopause.
Methods
The review analyzes the hormonal interactions that impact migraine and MS, emphasizing the role of estrogen in disease modulation across different reproductive stages. Following SANRA guideline an online search was conducted in PubMed, Scopus, WOS, and Embase, the used keywords included: “Migraine,” “multiple sclerosis,” “women,” “hormones,” “estrogen,” “menstrual cycle,” “pregnancy,” “postpartum,” “contraceptives,” “menopause,” “reproductive health,” “obstetrics,” and “gynecology.” The included articles were English peer-reviewed studies from the last 20 years that addressed MS or migrant in women of reproductive age group, we excluded case reports, editorials, and non -relevant studies. The included articles were assessed for quality, clinical relevance, and recency. Extracted data were synthesized to reflect how hormonal changes across diverse women's life cycle influence the disease pattern and management. The key finding was organized thematically and presented to support evidence-based integration into obstetrician-gynecologists' (OB-GYNs) care.
Results
Estrogen withdrawal during the perimenstrual and postpartum periods exacerbates both migraine and MS activity. Stable high estrogen levels during pregnancy provide neuroprotection and reduce MS disease activity. The review also highlights the need for individualized treatment strategies involving contraceptives, hormone replacement therapies (HRT), and assisted reproductive technologies (ART). Menopause leads to increased disease activity in both conditions due to declining estrogen levels.
Discussion
The current study highlights the integral role of estrogen fluctuation in modulating disease activity in both migraine and MS, with steady hormonal states present a protective effect. it underscores the necessity of a tailored hormone-informed care strategy and multidisciplinary collaboration to enhance women's quality of life as well as neurological and reproductive health.
Conclusion
Despite advances in understanding hormonal modulation of migraine and MS, clinical practice often lacks integrated care strategies, particularly between neurologists and OB-GYNs. Collaborative care is essential for optimizing disease management and reproductive health outcomes. Enhanced awareness, preconception counseling, medication safety assessment, and lifestyle interventions are crucial for supporting women navigating the complex intersection of neurological and reproductive health.
1. INTRODUCTION
Migraine and multiple sclerosis (MS) are prevalent and disabling neurological conditions that disproportionately affect women, particularly during their reproductive years. Migraine is recognized as one of the leading causes of disability worldwide, with a global prevalence estimated at approximately 15%, and a female-to-male ratio of roughly 3:1 [1, 2]. Similarly, MS, a chronic autoimmune demyelinating disease of the central nervous system, exhibits a striking female predominance, with incidence rates in women two to three times higher than in men [3]. This gender disparity is believed to be influenced in large part by sex hormones, particularly estrogen, which modulates immune function, neuronal excitability, and vascular reactivity, all of which are relevant to migraine and MS pathophysiology.
It is now well established that estrogen fluctuations during critical reproductive transitions such as puberty, menstruation, pregnancy, postpartum, and menopause can significantly modulate disease onset, activity, and symptom severity in both migraine and MS [4]. For instance, the exacerbation of migraine attacks during menstruation (menstrual migraine) [5], and the tendency for MS relapse rates to decrease during pregnancy [6], but increase postpartum period [7] are widely recognized phenomena. Moreover, hormonal contraceptives and hormone replacement therapy (HRT) may influence disease trajectories [8, 9], raising important considerations for reproductive health management in affected women. However, the precise mechanisms linking estrogen and reproductive milestones to the pathophysiology of migraine and MS remain incompletely understood and continue to be a subject of active investigation.
Despite growing research interest, the existing literature presents conflicting data on the extent and nature of the hormonal influence on both diseases. For migraine, some studies report a protective effect of pregnancy on migraine frequency [10], while others highlight increased susceptibility postpartum [11]. Similarly, while MS is widely considered to stabilize during pregnancy [12], reports of disease rebound postpartum and the potential impact of assisted reproductive technologies (ART) remain areas of ongoing debate [13]. Furthermore, while hormonal therapies such as combined oral contraceptives (COCs) may provide symptom relief for migraine [14], their use in women with MS raises concerns regarding potential thrombotic risks and disease activity modulation. This variability underscores the need for nuanced, individualized care strategies that balance neurological and reproductive considerations.
This narrative review aims to evaluates how endogenous and exogenous estrogen–progesterone states across key reproductive milestones menarche, menstrual-cycle phases, pregnancy, the postpartum/lactation period, perimenopause, and menopause and the use of hormonal contraception or hormone replacement therapy influence disease activity in migraine and multiple sclerosis, including migraine frequency, severity, and aura, as well as MS relapses, MRI activity, and disability (EDSS). Building on this appraisal, the article provides actionable, stage-specific guidance on contraception, preconception and pregnancy care, lactation, and peri-/postmenopausal hormone therapy for women with migraine and/or MS, outlining clear co-management pathways between neurology and obstetrics/gynecology.
2. METHODOLOGY
2.1. Review Design And Epistemological Stance
This narrative review adopts an interpretivist/subjectivist paradigm [15]. We synthesize evidence to illuminate how hormonal transitions across the female reproductive lifespan shape the clinical interplay between migraine and multiple sclerosis (MS), with an emphasis on implications for obstetric and gynecologic (OB-GYN) care. In keeping with narrative review traditions, we prioritize conceptual integration and contextual interpretation over exhaustive enumeration. The perspectives and disciplinary backgrounds of the authors informed question framing, the selection of seminal literature, and interpretation of findings; consequently, the insights are context- and time-bound and may vary across clinical settings and teams. We followed SANRA (Scale for the Assessment of Narrative Review Articles) to enhance transparency and relevance [16]. Registration of the review at Open Science Framework: http://osf.io/e7bkz.
2.2. Search Strategy And Information Sources
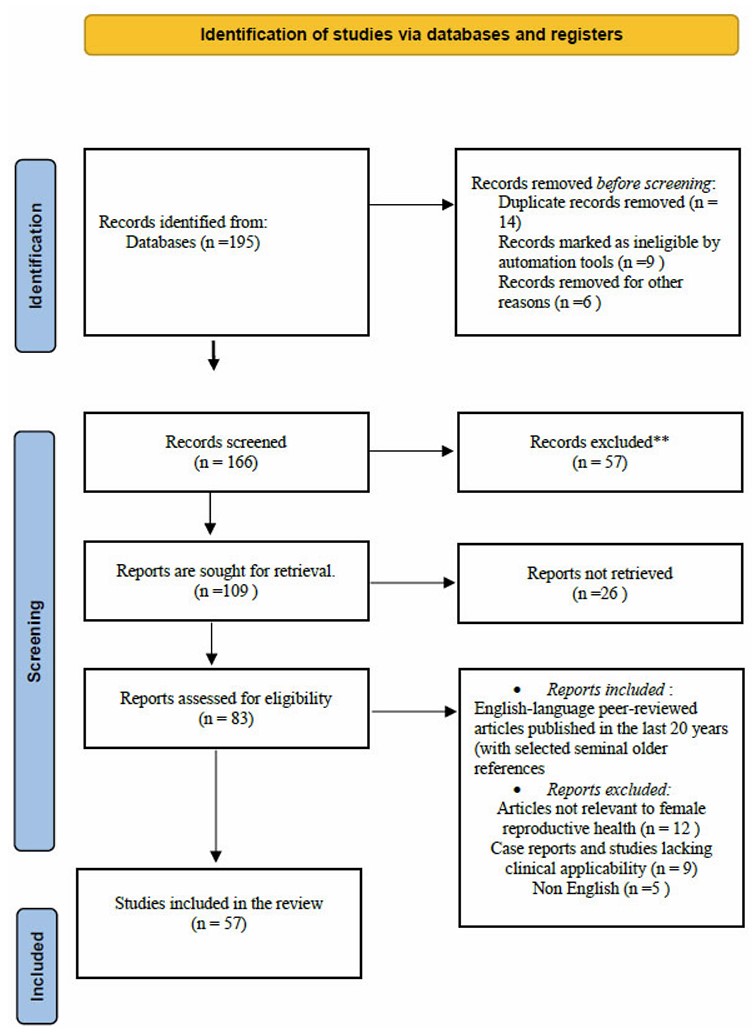
A purposeful, comprehensive search (January 2004–June 2025) was conducted in PubMed, Scopus, Web of Science, and Embase using controlled vocabulary and free-text terms related to migraine, MS, female reproductive health, and hormones (e.g., “migraine,” “multiple sclerosis,” “women,” “estrogen,” “menstrual cycle,” “pregnancy,” “postpartum,” “contraceptives,” “menopause,” “reproductive health,” “obstetrics,” “gynecology”). Reference lists of key publications and authoritative reviews were snowballed to identify additional influential articles. Search details and the selection pathway are summarized in Fig. (1).
3. RESULTS
3.1. Epidemiology and Gender Disparities
Migraine are approximately three times more prevalent in women than in men, with women experiencing longer attack durations and greater disability. This disparity emerges after puberty and persists through menopause, highlighting the role of sex hormones in disease manifestation [17]. Similarly, MS exhibits a higher incidence in women, with a female-to-male ratio of about 3:1. This difference is most pronounced during reproductive years and diminishes after menopause [18, 19].
3.2. Pathophysiological Links with Female Hormones
Hormonal fluctuations, particularly in estrogen levels, significantly impact both migraine and MS. In migraine, estrogen withdrawal is associated with increased attack frequency, as seen during menstruation and postpartum periods [20]. In MS, pregnancy, a state of elevated estrogen is linked to reduced relapse rates, suggesting a neuroprotective role of the hormone. Conversely, the postpartum drop in estrogen correlates with increased relapse risk [4].
3.3. Migraine and MS Across the Reproductive Timeline
The comparative analysis, as clarified in Table 1 highlights how hormonal fluctuations uniquely influence the clinical behavior of migraine and multiple sclerosis (MS) across key phases of the female reproductive lifespan. During puberty and the menstrual cycle, migraine symptoms often emerge or intensify due to estrogen withdrawal during the perimenstrual period [21]. In contrast, while early menarche has been associated with an increased risk of developing MS, the influence of cyclical hormonal changes on relapse activity in MS remains less clearly defined [22].
| Life stage | Hormone pattern | Clinical signs (typical) | Practical OB-GYN/Neuro takeaway | Key references |
|---|---|---|---|---|
| Puberty & menstrual cycle | Perimenstrual estrogen withdrawal | Increased attacks around menses; menstrual migraine; higher incidence post-puberty | Anticipatory management: consider short-term perimenstrual prophylaxis; avoid estrogen triggers when possible | MacGregor 2020 [57] |
| Preconception planning | Baseline endogenous hormones; medication rationalization | Most patients can optimize non-pharmacologic care; they need safe acute/preventive choices | Prefer acetaminophen first-line; consider NSAIDs/triptans with trimester-aware counseling; cautious preventive use (low-dose β-blocker, amitriptyline) | Ibrahim 2024 [43] |
| Pregnancy | High, relatively stable estrogen (↑ in 2nd–3rd trimesters) | Frequency often decreases, especially in the 2nd–3rd trimesters | Emphasize non-drug measures; use pregnancy-compatible acute meds if needed | Dumitru 2022 [23] |
| Postpartum | Rapid estrogen drop | Rebound/worsening attacks; breastfeeding may help | Encourage breastfeeding when feasible; plan acute therapy compatible with lactation | Tileshkumar 2023 [25] |
| Contraceptive use | Exogenous estrogen ± progestin | Estrogen-containing methods can exacerbate migraine, especially with aura | Prefer progestin-only or non-hormonal methods in aura; individualize choice | Tiranini 2023 [58] |
| Perimenopause & menopause | Estrogen fluctuation → decline | Perimenopausal worsening; variable postmenopausal course | Stabilize fluctuations; consider non-estrogen options; careful HRT consideration if indicated | Klimentova 2023 [59] |
| Fertility impact | No direct hormonal impairment from migraine | Fertility generally preserved; indirect effects via stress/meds | Pre-ART counseling focuses on trigger control and medication safety | Harpe 2022 [33] |
| Miscarriage/congenital risk | N/A (disease effect minimal) | No direct increase in risk | Reassure; focus on safe medication profiles | Phillips 2024 [35] |
| Effect of ART | Supraphysiologic estradiol; GnRH agents | No clear worsening; rare thrombotic events (e.g., CVST) reported | Coordinate with fertility team; monitor vascular risk; select migraine-safe regimens | Bakkensen 2022 [60]; Ersöz 2024 [40] |
| DMT/Hormonal medication safety* | N/A for DMTs (migraine) | Avoid known teratogens (e.g., valproate); some hormones may aggravate migraine | Choose pregnancy- and lactation-compatible acute/preventive therapies | Wells-Gatnik 2023 [37] |
| Pre-ART recommendations | Medication review before stimulation | Optimize control; rationalize acute/preventive plan | Document a safe rescue plan before ovarian stimulation | Crowe 2022 [38] |
| Post-ART monitoring | Post-stimulation/hormone normalization | Watch for rare CVST; symptom variability | Early neuro evaluation if atypical headache/red flags | Ersöz 2024 [40] |
| Lactation considerations | Relatively stable low estrogen during lactation | Continued migraine relief in some; compatible meds available | Acetaminophen, ibuprofen, and sumatriptan are considered compatible | Tileshkumar 2023 [25] |
*DMT = disease-modifying therapy (listed here only to clarify that DMT guidance applies to MS, not migraine).
| Life stage | Hormone pattern | Clinical signs (typical) | Practical OB-GYN/Neuro takeaway | Key references |
|---|---|---|---|---|
| Puberty & menstrual cycle | Post-pubertal estrogenization; peri-menses fluctuation | Earlier menarche linked to higher MS risk; menstrual influence on relapses unclear | Routine care; symptom diaries may help differentiate cyclical effects | Ucciferri 2022 [22] |
| Preconception planning | Baseline endogenous hormones; DMT planning | Best outcomes with stable disease 6–12 months pre-conception; vitamin D optimization | Discontinue most DMTs before conception unless high-risk; supplement vitamin D | Wang 2023 [6]; Graham 2024 [41]; Mansur 2022 [42] |
| Pregnancy | High, stable estrogen (peaks late pregnancy) | Relapse reduction—greatest in 3rd trimester (up to ~70%) | Reassure; plan postpartum strategy early | Dobson 2020 [61] |
| Postpartum | Abrupt estrogen decline | Relapse risk peaks at ~3–4 months postpartum | Early DMT resumption when indicated; breastfeeding may reduce relapses; monitor for preeclampsia when relevant | Anderson 2021 [62]; Krysko 2021 [63]; Villaverde-González 2022 [51] |
| Contraceptive use | Exogenous hormones | Minimal effect on progression overall | Choose a method based on individual risk, comorbidities, and DMT plan | Al-Khawajah 2023 [47] |
| Perimenopause & menopause | Estrogen fluctuation → decline | Earlier menopause is associated with earlier progression; estrogen loss may accelerate disability | Anticipate functional needs; discuss non-estrogen symptom control; consider bone health | Lorefice 2023 [30] |
| Fertility impact | Disease activity and treatment context | Fertility is generally preserved; subtle ovarian reserve reduction is possible in active disease | Coordinate with fertility specialists; assess ovarian reserve when disease is active | Arabipoor 2024 [31]; Carbone 2023 [32] |
| Miscarriage/congenital risk | N/A (disease effect minimal) | No direct increase in risk | Standard obstetric risk counseling; align DMT timing | Andersen 2021 [34] |
| Effect of ART | Supraphysiologic estradiol; GnRH agents | Early data: ART does not increase relapse risk; recent data: lower risk with DMT maintenance | Favor continuation of compatible first-line DMTs during ART when appropriate | Bakkensen 2022 [60]; Range 2023 [64] |
| DMT during ART/pregnancy | Balancing fetal safety and maternal control | Interferons/glatiramer are preferred in ART/pregnancy contexts when needed | Select pregnancy-compatible DMTs; plan postpartum escalation if indicated | Stamatellos 2023 [36] |
| Pre-ART recommendations | Disease stability assessment | Stabilize disease; evaluate ovarian reserve | Multidisciplinary planning with timelines for DMT holds/resumption | Sparaco 2023 [13] |
| Post-ART monitoring | Post-stimulation period | Neurological monitoring for 3–6 months | Early follow-up to detect relapse/activity | Rankin 2018 [39] |
| Lactation considerations | Lactational low estrogen | Breastfeeding may reduce postpartum relapses; some DMTs are compatible | Prefer glatiramer acetate/interferon-β; avoid monoclonals during breastfeeding | Krysko 2021 [63] |
In pregnancy, both conditions generally show improvement. Migraine frequency tends to decrease, particularly in the second and third trimesters, mirroring the rise in stable estrogen levels [23]. Similarly, MS relapse rates significantly decline—up to 70% in the third trimester, suggesting a protective immunomodulatory effect of pregnancy-related hormones [24].
However, the postpartum period introduces a sharp decline in estrogen levels, often triggering a resurgence in symptoms. Migraine attacks frequently return or worsen [25], while MS patients face an elevated relapse risk, peaking around 3–4 months after delivery [7]. Some evidence suggests that breastfeeding, by maintaining hormonal stability, may help reduce the severity or timing of postpartum relapses in MS [26].
Regarding contraceptive use, estrogen-containing hormonal contraceptives can exacerbate migraines, particularly in women who experience migraine with aura, and may increase the risk of cerebrovascular events [27]. MS, however, does not appear to be significantly influenced by contraceptive hormones, although contraceptive choice should still be personalized based on the individual’s clinical profile and comorbidities [28].
Finally, during perimenopause and menopause, the decline and irregularity of estrogen can aggravate migraine frequency and severity. Some women may experience symptom relief post menopause [29]. In MS, early menopause has been correlated with an earlier transition to progressive disease and greater disability, likely linked to the loss of estrogen’s neuroprotective effects [30]. The details of Migraine and MS across the Female Reproductive Timeline are shown in Fig. (2)
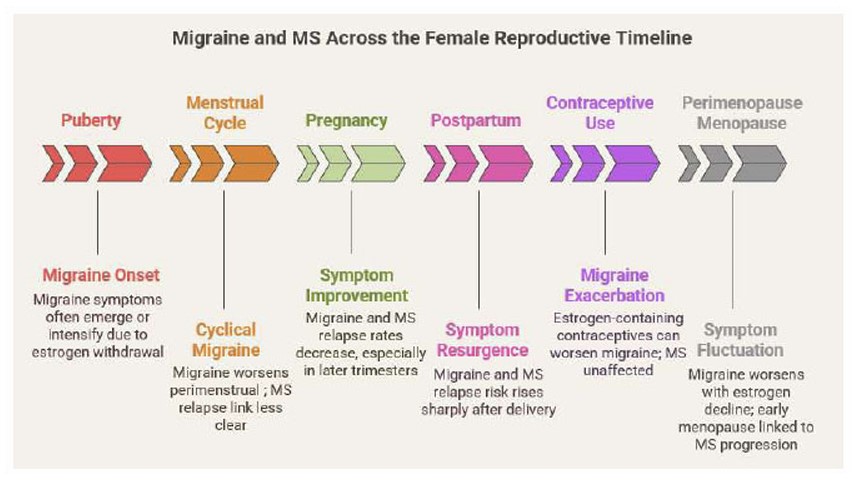
Migraine and MS Across the Female Reproductive Timeline.
3.4. Implications for Fertility and Assisted Reproductive Technology
Fertility outcomes in both multiple sclerosis (MS) and migraine are generally reassuring. In MS, most women maintain normal fertility [31], although subtle reductions in ovarian reserve may occur, especially in cases of high disease activity [32]. In contrast, migraine has no known direct impact on fertility, though stress and medication use may exert indirect effects [33].
Neither MS [34] nor Migraine [35] has been definitively linked to increased risks of miscarriage or congenital anomalies, reinforcing that pregnancy is generally safe in terms of fetal outcomes.
Regarding assisted reproductive technology (ART), earlier studies in MS raised concerns about a heightened relapse risk following hormonal stimulation. However, more recent evidence suggests that maintaining disease-modifying therapies (DMTs) significantly lowers relapse rates, making ART a safer option when managed appropriately [13]. For migraine, limited data exist on ART-specific effects, but no consistent evidence points to worsened outcomes following such interventions.
Medication management is critical in both populations. In MS, first-line DMTs such as interferons and glatiramer acetate are preferred during ART due to their favorable safety profiles [36]. For migraine, teratogenic medications like valproate should be avoided, and hormonal treatments may exacerbate symptoms in hormone-sensitive patients [37].
Pre-ART planning for MS includes disease stability assessment and ovarian reserve evaluation [13], whereas for migraine, the focus is on ensuring medication safety and symptom control [38]. Post-ART monitoring is especially important in MS, with neurological follow-up advised for 3–6 months [39]. Notably, cerebral venous sinus thrombosis has been reported following IVF, particularly in women with a history of migraine and underlying thrombophilia risk factors [40].
Multidisciplinary coordination is essential in both conditions. MS management requires collaboration between neurologists and fertility specialists, whereas in migraines, OB-GYN consultation is recommended to guide safe reproductive planning and medication adjustments.
3.5. Management Considerations During Pregnancy and Lactation
The management of migraine and MS during pregnancy requires careful consideration of medication safety and disease control. Many disease-modifying therapies (DMTs) for MS are contraindicated during pregnancy due to potential teratogenic effects [6, 41]. Conversely, certain supplements including vitamin D, may be safely continued or initiated during pregnancy and lactation [42]. For migraines, non-pharmacological treatments are preferred, including lifestyle modifications and avoiding known triggers. When medication is necessary, acetaminophen is the first-line treatment, with nonsteroidal anti-inflammatory drugs and triptans regarded as secondary options, though trimester-specific risks limit their use. Preventive treatments, if required, may include low doses of β-blockers or amitriptyline but should be used cautiously [43].
3.6. Clinical Guidance for OB-GYNs in the Management of Migraine and Multiple Sclerosis
Managing migraine and multiple sclerosis (MS) in women requires a collaborative and tailored approach, particularly during reproductive milestones such as preconception, pregnancy, and postpartum. OB-GYNs play a pivotal role in multidisciplinary coordination, especially when managing treatment safety and hormonal influences.
For hormonal management, non-hormonal contraceptives like copper IUDs are often preferred for women with migraine with aura due to stroke risk [44], meanwhile hormone therapies in MS may be beneficial when tailored, particularly with agents like high-dose estriol that demonstrate anti-inflammatory effects [45].
Preconception counseling is essential for both groups. Preconception counseling is crucial for women with migraine, as it can help optimize pregnancy outcomes and manage risks While migraine requires review of medication safety and nutritional support (e.g., folic acid, vitamin D) as studies have shown that pre-pregnancy migraine is associated with increased risks of adverse pregnancy outcomes, including preterm delivery, gestational hypertension, and preeclampsia [46], MS demands thorough assessment of disease stability and possible adjustments in DMTs before conception [47], Patients exposed to second/third-line DMTs like natalizumab and fingolimod before conception are at higher risk of MS reactivation during pregnancy [48]. ART procedures necessitate careful neurologic monitoring in MS [49].
In the postpartum period, hormonal shifts may trigger migraine exacerbations, best managed with non-pharmacologic options such as acupuncture [50]. MS patients face a high risk of relapse during this time, requiring prompt DMT resumption and blood pressure monitoring for potential preeclampsia [51].
Finally, Lifestyle modifications such as regular physical activity, sufficient sleep, and a heart-healthy diet offer benefits in managing both conditions [52, 53]. However, particular attention should be given to cognitive function in migraine patients, as the relationship between migraine and cognitive impairment is bidirectional. Migraines can lead to cognitive dysfunction, while cognitive decline may, in turn, aggravate migraine symptoms [54]. In patients with multiple sclerosis (MS), cognitive impairment is a major concern, affecting up to 65% and significantly diminishing quality of life [55]. Additionally, caution is warranted with medications like topiramate, commonly used in Migraine and MS, as they may further impair cognitive performance [56].
Hormone-linked clinical patterns and management considerations are summarized separately for migraine Table 1A and MS Table 1B.
4. DISCUSSION
Migraine and multiple sclerosis (MS) are two chronic neurological conditions that disproportionately affect women, with their activity and clinical course intricately modulated by fluctuations in sex hormones, particularly estrogen across the female reproductive lifespan. An accumulating body of literature supports that hormonal transitions such as puberty, pregnancy, and menopause significantly influence disease behavior in both disorders, yet clinical practice has yet to fully integrate these insights into routine care strategies.
Estrogen withdrawal has been strongly linked to increased migraine frequency, explaining the prevalence and intensity of attacks during the perimenstrual and postpartum periods that were found in the review study of MacGregor et al. [57], that details how estrogen fluctuations act as a potent migraine trigger, especially in the context of hormonally sensitive phenotypes such as menstrual migraine. On the other hand, Lorefice et al. [30] have shown that estrogen exerts a neuroprotective effect in MS, particularly evident during pregnancy when relapse rates drop by up to 70%, a trend also emphasized by Dobson et al. [61]. Conversely, the postpartum period, with its abrupt estrogen decline, is marked by a spike in MS relapses, as confirmed by Schubert et al. [7] and Anderson et al. [62], highlighting a phase of substantial clinical vulnerability.
Despite robust research findings, the integration of hormonal considerations into the clinical management of women with these conditions remains limited, especially within obstetrics and gynecology. The current review underscores a gap in interdisciplinary collaboration, whereby OB-GYNs often lack specialized neurological knowledge critical to guiding reproductive planning, contraceptive choice, and pregnancy care for women with migraine and MS. This gap can lead to missed opportunities for anticipatory counseling and may compromise patient safety, particularly when managing teratogenic medications or navigating high-risk reproductive interventions.
The literature reveals both shared and distinct aspects in how migraine and MS interact with reproductive health. For instance, fertility remains largely preserved in both conditions. However, MS may subtly reduce ovarian reserve in active disease, as demonstrated by Carbone et al. [32], whereas migraine's influence is more indirect, potentially mediated by stress or pharmacotherapy Harpe et al. [33]. Assisted reproductive technology (ART) has emerged as another critical intersection. Early studies suggested a heightened relapse risk in MS following hormonal stimulation, but more recent data, such as that by Range et al. [64], and Bakkensen et al. [60] support the safety of ART when disease-modifying therapies (DMTs) are appropriately maintained. For migraine, the data remain limited, but Ersöz et al. [40] documented a case of cerebral venous sinus thrombosis post-IVF, warranting vigilance in high-risk patients.
Medication safety is another cornerstone. In MS, first-line DMTs like interferons and glatiramer acetate have demonstrated relative safety during ART and early pregnancy Stamatellos et al. [36]. In contrast, certain migraine preventives, such as valproate, are known teratogens and must be avoided in reproductive-aged women Wells-Gatnik et al. [37]. Hormonal contraceptives also pose divergent considerations: while they may exacerbate migraine with aura and increase stroke risk Tiranini et al. [58], they appear to have minimal impact on MS progression Al-Khawajah et al. [47].
Clinicians should also be aware of how menopause affects disease trajectories. Perimenopausal migraine often worsens due to hormonal instability, but has a chance of improvement. postmenopause, Klimentova et al. [59], whereas MS patients may experience accelerated disability following menopause due to the loss of estrogen’s neuroprotective modulation, as described by Lorefice et al. [30]. These findings further underscore the need for tailored, hormone-aware strategies throughout a woman’s life.
The strength of this review lies in its comprehensive synthesis of the literature across multiple reproductive stages, emphasizing on practical clinical intersections between OB-GYN and neurology. It offers evidence-based guidance not only for managing acute disease but also for navigating fertility, pregnancy, ART, and lactation, all from a multidisciplinary lens. It contextualizes complex hormonal physiology within real-world management challenges and suggests how best to align neurologic safety with reproductive goals.
Nevertheless, limitations exist. As a narrative review, this study lacks the methodological rigor of systematic reviews or meta-analyses and is subject to selection bias in article inclusion. While the literature cited is current and comprehensive, some areas, such as ART outcomes in migraine patients or the cognitive effects of specific medications, remain underexplored and warrant further research.
Based on the findings, it is strongly recommended that OB-GYNs receive more targeted training in neurological comorbidities such as MS and migraine. Clinical pathways should be redesigned to promote seamless integration between neurology, reproductive medicine, and primary care. Furthermore, shared decision-making models must be employed, ensuring that women are fully informed about medication risks, hormonal implications, and lifestyle interventions tailored to their specific condition. Non-pharmacologic strategies such as stress management, regular sleep, and diet should be emphasized as foundational tools for both prevention and symptom control Mallardo et al. [53].
Future studies should address how the female reproductive life cycle affects therapeutic strategies. Despite the growing interest in gender specific medicine, there is still a paucity of evidence addressing the hormonal fluctuations across the life span. There are encouraging results suggesting that probiotic intervention can modulate the neuroinflammatory pathways in MS [65, 66].
Other studies have looked into reducing oxidative stress with the use of inulin supplements and showed a promising effect on reducing migraine symptoms among women [67, 68].
Non-pharmacological approaches are also worthy of mentioning, such as yoga, meditation, and cognitive behavioral therapy. This intervention has shown efficacy in reducing fatigue, anxiety, and improving QOL among MS women [69].
However, few studies have examined these interventions through a woman's reproductive lens. Future studies should adopt hormone-aware trial designs that account for reproductive life cycle phase for a more personalized therapeutic strategy to improve women's long-term neurological outcome. To advance gender sensitive neurology, there is a growing need for a high-quality stratified RCT that investigates gut-brain axis intervention in women suffering from migraine, since the current evidence remains limited and tentative [70].
In light of the critical role of estrogen in MS and migraine pathophysiology, it is vital to design studies that stratify women's reproductive phase to verify phase-specific responses [71].
Combining more than one therapeutic intervention is also worthy of exploration, i.e., inulin with a non-pharmacological approach, which offers the advantage of a synergistic effect in hormonally sensitive cases. Embracing a multimodal reproductive life stage-informed approach will allow effective, holistic care for women with MS and migraine [72, 73].
4.1. Clinical Integration and Care Pathways
Effective care for migraine and multiple sclerosis (MS) across the reproductive lifespan hinges on structured collaboration between neurology and obstetrics/gynecology (OB/GYN). A unified referral route with agreed indications—such as hormonally sensitive migraine at menarche, pre-conception counseling in MS, contraception decisions in migraine with aura, trimester-specific medication review, postpartum relapse planning, and escalation at perimenopause—facilitates timely access. Shared electronic health records should include a concise “Reproductive–Neurologic Summary” (phenotype, disease activity trend, current/held medicines including DMTs, vascular risks, fertility/contraception plan, and the next hormone-stage milestone), while synchronized follow-ups anchor care at pivotal time-points (menarche/late adolescence, pre-conception, each trimester, early postpartum, and perimenopause). Contraceptive decisions are optimized by first classifying neurologic status (migraine with vs without aura; MS on or off DMTs, disability, and vascular comorbidity) and then balancing efficacy and vascular risk: combined estrogen methods are generally avoided in migraine with aura or high vascular risk and used selectively without aura; progestin-only methods are preferred when minimizing estrogen exposure and are typically acceptable in MS; non-hormonal options remain suitable at any stage. Pregnancy planning benefits from explicit checklists that confirm disease stability, specify DMT continuation/hold and washout timelines, adjust migraine preventives away from teratogens, and address vaccinations, thrombotic risk, and assisted reproduction. Antenatal care emphasizes diary-based monitoring, trimester-appropriate acute therapies, conservative MRI policies, and delivery planning, with peripartum protocols for postpartum relapse prevention, DMT resumption, and lactation-compatible regimens.
Postpartum and lactation pathways prioritize non-pharmacologic strategies for migraine, use lactation-compatible acute options where needed, and schedule MS reviews at ~6 weeks and 3–4 months to consider early DMT re-initiation and monitor hypertensive risk. Perimenopause and menopause management relies on individualized risk–benefit assessment that incorporates migraine subtype, MS status, vascular risks, and bone health; when indicated, low-dose transdermal estradiol with stable delivery is favored, with careful monitoring for aura change and MS activity and predefined thresholds for therapy adjustment. Patient-facing materials—plain-language stage-specific handouts and decision aids for contraception, pregnancy timelines with DMT checkpoints, and HRT risk–benefit grids—support shared decision-making; symptom diaries for headaches and relapses can be integrated through the patient portal. Service quality is tracked via pragmatic metrics, including postpartum MS relapse rate within six months and time-to-DMT resumption, migraine days per month during perimenopause and early postpartum, contraceptive method appropriateness in migraine with aura, timeliness of synchronized visits, documentation of lactation compatibility at discharge, presence of shared neurology–OB/GYN notes at key milestones, and patient-reported outcomes (satisfaction, decisional conflict, and health-related quality of life). These pathways translate hormone-informed evidence into coordinated, stage-specific practice that is auditable and adaptable.
CONCLUSION
Hormonal fluctuations across the reproductive lifespan meaningfully modulate disease activity in migraine and multiple sclerosis, whereas more stable hormonal states often confer relative protection. Translating this evidence into care requires coordinated neurology–OB/GYN pathways that align contraception choices, pregnancy and postpartum planning, and peri-/postmenopausal hormone therapy with neurologic risk. Implementing the proposed co-management templates, decision grids, and stage-specific checklists can improve safety, symptom control, and patient experience for women navigating overlapping reproductive and neurological needs. Ongoing research should refine risk stratification and evaluate these pathways using standardized quality metrics.
AUTHORS' CONTRIBUTIONS
The authors confirm contribution to the paper as follows: M.T., E.A.M., W.N.M.H., Study conception and design; M.T.: Data collection; E.A.M., M.T.: Analysis and interpretation of results; W.N.M.H.: Draft manuscript;. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| MS | = Multiple Sclerosis |
| HRT | = Hormone Replacement Therapies |
| ART | = Assisted Reproductive Technologies |
| COCs | = Combined Oral Contraceptives |
| OB-GYNs | = Obstetrician-gynecologists |
| DMTs | = Disease-Modifying Therapies |
ACKNOWLEDGEMENTS
Declared none.