All published articles of this journal are available on ScienceDirect.
Tuberculosis in Low- and Middle-income Countries: A Critical Analysis of Health System Barriers and the Promise of Artificial Intelligence
Abstract
Introduction
The burden of Tuberculosis (TB) is disproportionately high in low- and middle-income countries (LMICs), where structural health-system constraints and social disparities delay diagnoses and undermine treatment outcomes. This review analyzed evidence across five high-burden LMICs (India, Indonesia, Nigeria, the Philippines, Pakistan). It appraised the emerging role of Artificial Intelligence (AI) in TB diagnosis, monitoring treatment adherence, and assessing outcomes.
Methods
The review followed PRISMA guidelines and conducted a Johns Hopkins Evidence-Based Practice appraisal of peer-reviewed studies, national program reports, and selected grey literature (January 2015–December 2025). Published data on epidemiology, health financing, service integration, diagnostics, and digital/AI-powered interventions were analyzed and summarized into descriptive themes. Classic TB publications date back to 2005.
Results
India had the largest absolute TB and multidrug-resistant TB (MDR/RR-TB) burden, while Nigeria recorded the highest mortality and TB/HIV co-infection prevalence. Case detection rates ranged from ~64% to 79%, and treatment success rates ranged from ~74% to 86% across countries. Domestic funding for TB control programs accounted for 80% or more of national TB budgets in India. Nigeria, Pakistan, and the Philippines reported that less than 50% of their TB control programs' funding came from local sources, thereby relying on donor funding to cover budget deficits. AI-driven chest radiography and technology for monitoring TB treatment adherence and outcomes showed promise. Researchers in pilot studies found that AI-assisted TB diagnoses, treatment access, and outcome monitoring were hindered by poor infrastructure, inadequate and untrained TB healthcare staffing, scalability, and sustainability constraints. Additionally, social disparities, environmental factors, and stigma adversely affect early diagnosis and management of TB among rural dwellers, women, children, and hard-to-reach communities.
Discussion
National TB funding deficits and fragmented national health systems and insurance schemes resulted in weak surveillance and poor TB/HIV care integration treatment outcomes. Although AI applications can augment TB screening and monitoring of treatment adherence and outcomes, scalability, equity, and the ethical and transparent use of AI technology were hampered mainly by funding deficits and poor digital infrastructure. Research validating the impact of AI on national TB programs was primarily conducted through pilot investigations.
Conclusion
Progress toward End TB targets will require robust domestic funding, integration of TB control programs into primary and national insurance schemes, and a phased, scalable, and context-appropriate approach to AI deployment. TB control programs in LMICs will benefit from incorporating AI tools within broader health-system reforms to ensure equitable, sustainable TB control.
1. INTRODUCTION
Tuberculosis (TB) is a persistent and deadly global epidemic. In 2022, the World Health Organization (WHO) estimated that 10.8 million people had TB. An estimated 1.3 million HIV-negative TB patients and 167,000 HIV positive TB patient deaths were recorded in 2022 [1]. Despite decades of global interventions and commitments, TB still ranks as the second-leading cause of communicable disease deaths after COVID-19 (1). LMICs bear a disproportionate burden of TB. Approximately 86% of global TB patients live in LMICs, with five countries-India, Indonesia, the Philippines, Pakistan, and Nigeria accounting for over 55% global TB cases (2). India reported about 2.8 million TB cases in 2022, representing 26% of the global TB burden [2]. In 2022, an estimated 467,000 people were infected with TB in Sub-Saharan Africa, calling attention to the crisis in the region [3]. High HIV co-infection, widespread MDR-TB, and systemic healthcare access disparities in LMICs frequently compound tuberculosis.
Global TB research and funding continue to be disproportionately driven by High-Income Countries (HICs). Consequently, clinical guidelines, diagnostics, and therapeutic strategies are often developed without the input of LMIC governments or health systems [4]. Notable gaps in research on pediatric TB, extrapulmonary TB, and gender-specific barriers to access are persistent, all of which are more prevalent and complex in LMICs [5-7]. Additionally, the weakness of health systems across many LMICs, fragmented health systems and health insurance, low patient-to-healthcare worker ratios, and inadequate laboratory infrastructure continue to exacerbate the situation. Inconsistent integration of TB services into primary healthcare is militating against timely diagnosis and treatment [8,9]. Poverty, food insecurity, stigma, and social disparities are critical social determinants that delay treatment-seeking and exacerbate disease progression among women, children, and rural dwellers [10,11].
Advances in Artificial Intelligence (AI) and the use of digital health technologies offer opportunities for enhanced tuberculosis (TB) control. The CAD4TB, an AI-driven chest radiographic interpretation app, and mobile-based adherence technologies such as 99DOTS and Video-Observed Therapy (VOT) improved TB diagnosis and treatment monitoring in pilot studies [12, 13]. However, scalability challenges, concerns about transparent use, poor infrastructure, and ethical concerns around data privacy are barriers to widespread adoption in LMICs [14–16]. The enumerated challenges justify the need for further research and a comprehensive evaluation of TB control programs in LMICs. Further research must transcend biomedical metrics and encompass rigorous scrutiny of the systems, social disparities, and the prospects for large-scale deployment of AI technology. This review sought to scrutinize the key health system barriers to Tuberculosis (TB) control in five high-burden low- and middle-income countries (LMICs)- India, Indonesia, Nigeria, the Philippines, and Pakistan- and to evaluate the prospects of current artificial intelligence (AI) technology in augmenting TB diagnoses and treatment. The research questions for this review are (1) What are the primary health system and operational weaknesses within national TB programs that contribute to persistent incidence and mortality in LMICs? (2) How has the implementation of digital and AI technology influenced early detection, treatment monitoring, and data management? (3) What is the impact of AI integration on national policy and health system strategies for TB prevention, treatment, and control? These questions were the guiding framework for the search strategy, data analyses, discussion, and conclusions of the review.
2. METHODS
2.1. Review Design
The authors used an integrative review methodology according to the 2020 guidelines of the Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). This approach was selected to synthesize data across empirical, theoretical, and grey literature sources. The review also applied the Johns Hopkins Evidence-Based Practice (JHEBP) model to appraise the evidence critically, ensure methodological rigor, and assess relevance to health systems in low- and middle-income countries (LMICs).
2.2. Search Strategy
The authors conducted a comprehensive literature search of the following databases: PubMed, Scopus, and the WHO Global Index Medicus, covering the period from January 2015 to December 2025. Search terms included combinations of controlled vocabulary (MeSH) and free-text keywords related to “Tuberculosis,” “Low- and Middle-Income Countries,” “AI in TB,” “TB Surveillance,” “MDR-TB,” “TB/HIV Co-infection,” and “Health Systems Strengthening.” Boolean operators (“AND”, “OR”) and truncation were applied to refine results.
The authors searched the official publication sites of the WHO, Stop TB Partnership, and national TB control programs of India, Indonesia, Nigeria, the Philippines, and Pakistan for grey literature. Duplicates were removed manually using EndNote X9. Full texts were retrieved for articles meeting the eligibility criteria based on title and abstract screening.
2.3. Inclusion and Exclusion Criteria
2.3.1. Inclusion Criteria
• Peer-reviewed studies, government reports, and NGO publications
• Studies focused on TB incidence, mortality, management, or technology in LMICs
• Research involving health systems, social determinants, AI tools, or implementation challenges
• Studies involving populations in high-burden LMICs, especially India, Nigeria, Indonesia, the Philippines, and Pakistan
2.4. Study Selection and PRISMA Flow
We imported Data from the literature search into EndNote for duplicate removal, and two reviewers independently screened titles and abstracts. The reviewers independently evaluated full texts for relevance and quality and included eligible publications by consensus. A PRISMA flow diagram is presented in the results section to illustrate the selection process, including records identified, screened, assessed for eligibility, and included in the synthesis. The screening and study selection process is summarized in Fig. (1).
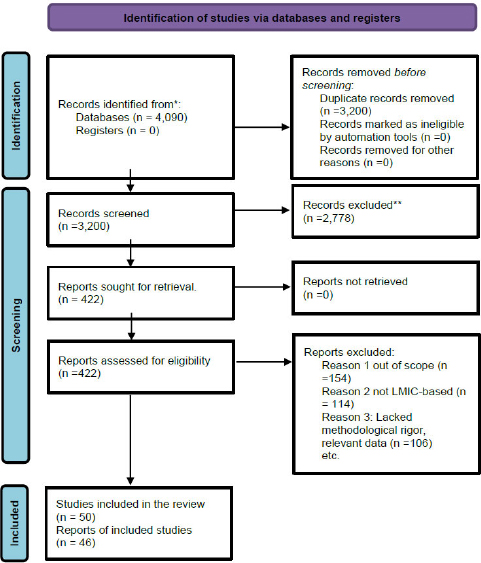
PRISMA 2020 study selection flow diagram for included evidence sources.
2.5. Methodological Limitations
Though the authors complied with PRISMA guidelines and conducted a structured critical appraisal of the selected publication, this review has limitations. Selection bias may persist due to the 2015 start date for most of the cited articles and the English-language focus. The inclusion of heterogeneous sources (peer-reviewed studies, national reports, and grey literature) may limit comparability. The authors did not conduct a formal inter-rater reliability test, and publication selection was determined by discussion and consensus; as a result, the reviewed article selection process may be subjective and biased. However, the authors sought to minimize the selection bias by using the Johns Hopkins Evidence-Based Practice (EBP) Research Evidence Appraisal Tool.
2.6. Quality Appraisal
The authors used the Johns Hopkins Evidence-Based Practice (EBP) Research Evidence Appraisal Tool to appraise the methodological quality of the peer-reviewed studies. This tool grades evidence from Level I (experimental) to Level V (non-research evidence) and rates quality as high, good, or low based on study design, sample adequacy, rigor, and relevance to practice. Grey literature from the WHO or national TB program reports was evaluated using a tailored version of the AACODS checklist (Authority, Accuracy, Coverage, Objectivity, Date, and Significance).
2.7. Data Extraction and Synthesis
The authors created a standardized data extraction form to categorize information, including author(s), year, country of study, study objectives, methods, population, TB-related outcomes (incidence, prevalence, mortality), health system characteristics, social determinants, and technological interventions. The authors used A mixed-method synthesis strategy. Quantitative data (e.g., TB rates and funding allocations) were summarized using descriptive statistics. Qualitative data (e.g., barriers to care, technology feasibility) were thematically analyzed and integrated narratively to identify commonalities, gaps, and implementation challenges.
3. RESULTS
3.1. Overview of Included Studies
The authors performed a comprehensive and rigorous, integrative review of 46 publications. These included 32 peer-reviewed journal articles, National Tuberculosis Program (NTP) reports, publications from reputable organizations such as the Stop TB Partnership and Médecins Sans Frontières, and four academic white papers. The literature in this review met the stringent methodological quality standards set by the Johns Hopkins Evidence-Based Practice (EBP) appraisal tool, a widely respected tool in public health research. The studies represented a broad spectrum of empirical designs, including cohort studies, implementation evaluations, epidemiological surveys, and systems analyses, spanning the period from January 2015 to December 2025.
3.2. Epidemiological Overview: Comparative Country-level Analysis
The burden of tuberculosis remains disproportionately high in LMICs. India accounts for the most significant global TB burden, with 199 cases per 100,000 population and 32 deaths per 100,000 in 2022. Indonesia reported 354 cases per 100,000 and 34 deaths per 100,000. Nigeria recorded 323 per 100,000 cases and a higher mortality (46 per 100,000). Nigeria’s high mortality rates are attributed to diagnostic delays and the compounding effect of TB/HIV co-infection. The Philippines recorded 650 cases per 100,000, the highest among the five countries, and 70 deaths per 100,000. Pakistan reported 275 cases per 100,000 and 42 deaths per 100,000. Together, the five countries accounted for more than half of all global TB cases in 2022. Figure (2) highlighting the geographic concentration of TB morbidity and mortality in LMIC settings [1, 2].
3.3. Summary Observations
1. Incidence: The Philippines and Nigeria had the highest TB incidence per capita.
2. Mortality: Nigeria had the highest TB-related deaths, high co-infection with HIV, and a weak diagnostic infrastructure.
3. Drug-Resistant TB: India has the highest absolute number of MDR-TB cases, but all five countries face growing burdens.
4. Detection and Success Rates: Despite high treatment success rates in Nigeria and Indonesia, low detection rates and missed diagnoses are common
5. There are persistent funding deficits in all five countries. Funding gaps, often filled by external donors, underscore the urgent need for increased local investment in TB management.
3.4. Health System Capacity and Integration
3.4.1. Case Detection and TB/HIV Co-infection
In 2022, India achieved the highest case detection rate among the five countries at 79%. Indonesia was second at 70%, then Nigeria at 67%, the Philippines (65%), and Pakistan (64%). These achievements are commendable, yet none of the countries met the End TB Strategy's target of≥90% detection, echoing the need for enhanced case detection and management of TB/HIV co-infection to help improve treatment outcomes and reduce mortality rates. HIV/TB co-infection is a major threat to TB control, particularly in sub-Saharan Africa, amplified by Nigeria’s high TB/HIV co-infection burden among the five countries. The rate of TB/HIV co-infection in Nigeria 21% compared to 3.5% in India and 3.0% in Indonesia [1,4,5]. The absence of integrated TB and HIV/AIDS control programs and limited point-of-care diagnostics contributes to late presentation and poor outcomes [17,18]. Conversely, co-infection rates are relatively low in India (3.5%), Indonesia (3.0%), the Philippines (3.7%), and Pakistan (2.9%) (1, 2, 4, 5). These comparative performance differences are illustrated in Fig. (3).
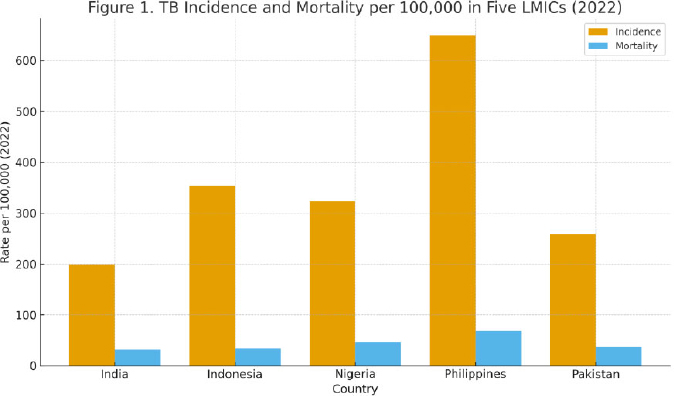
Tuberculosis incidence and mortality per 100,000 population in five high-burden LMICs (2022). Incidence and mortality estimates based on the WHO Global Tuberculosis Report 2023.
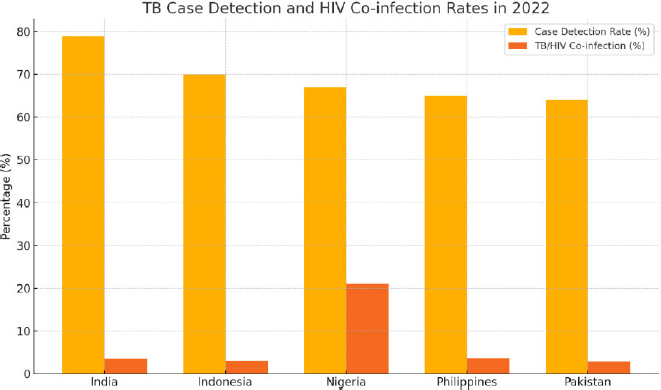
TB case detection and TB/HIV co-infection rates in five high-burden LMICs (2022). Case detection is the percentage of estimated TB cases that were diagnosed and reported. At the same time, TB/HIV co-infection represents the proportion of TB cases occurring in individuals with HIV infection-data. (Source: WHO Global Tuberculosis Report 2023).
3.4.2. Health System Integration
India and Indonesia carried out significant vertical and horizontal integration of TB and HIV co-infection services. India’s Nikshay platform links TB and HIV data at the facility level and coordinates with private providers to facilitate notifications and referrals [2]. Nigeria and Pakistan report weak integration, particularly in rural and faith-based facilities [3, 6]. The Philippines is piloting decentralization efforts, which are adversely affected by funding challenges and donor reliance [5].
3.4.3. Human Resources and Diagnostics
The five countries report serious workforce and diagnostic gaps, with most National Tuberculosis Programs (NTPs) facing recurrent shortages of trained TB nurses, laboratory technicians, and Community Health Workers (CHWs), particularly in rural districts [19, 20]. Staff attrition, low remuneration, and insufficient workforce incentives impede consistent diagnostic quality [21]. GeneXpert MTB/RIF deployment coverage is not consistent. India and Indonesia report broad deployment (>4,000 and >1,500 instruments, respectively). Still, Nigeria and the Philippines rely heavily on smear microscopy for peripheral diagnosis [22-24]. In several rural communities, a single GeneXpert machine may serve populations exceeding one million, contributing to diagnostic delays and under-detection of multidrug-resistant TB (MDR-TB) [25, 26]. Urban centers generally leverage GeneXpert technology for faster turnaround times and higher detection rates, reflecting disparities in laboratory density, connectivity, and maintenance infrastructure [27]. The social disparities highlight the critical need for equitable workforce investment and decentralized diagnostic capacity to achieve timely case identification and treatment initiation [28].
3.4.4. Surveillance Systems
Digital TB surveillance is becoming more common across all five countries. India’s Nikshay system is the most advanced and fully integrated nationwide. Pakistan’s ETR and the Philippines’ iDOTS are partially paper-based in rural areas. Nigeria’s TB reporting is predominantly vertical and donor-driven, with inconsistent subnational reporting (3, 4, 6).
3.4.5. TB Financing and Donor Dependence in High-burden LMICs
In 2022, local national TB budgets covered less than 50% program costs: Pakistan (40%), the Philippines (37%), and Nigeria (32%) [28, 29]. Significant reliance on external donor funding, primarily from the Global Fund and USAID, continues to stagnate progress and sustainability efforts. Without robust, sustained, and reliable domestic funding and integration into national health insurance schemes, TB control programs remain vulnerable to disruptions [29, 30].
3.4.6. Drug-resistant TB and Treatment Outcomes
India had the highest number of MDR-TB cases (132,000) in 2022, followed by Indonesia (25,000), Nigeria (21,000), the Philippines (20,000), and Pakistan (15,000) (1). Treatment success rates were Nigeria (86%), Indonesia (84%), India (82%), Pakistan (79%), and the Philippines (74%) (2-6).
3.5. Technology, Innovation, and AI in TB Management in LMICs
3.5.1. AI-Enhanced Radiographic Screening
Artificial intelligence-assisted chest radiography is a promising adjunct to large-scale TB screening in resource-limited settings. Computer-aided detection systems such as CAD4TB and qXR have demonstrated diagnostic accuracy comparable to that of radiologists in multicenter pilot evaluations [29, 30]. Researchers in India and South Africa found high CAD4TB sensitivity ranging from 94% to 96% and specificity ranging from 77% to 83% for bacteriologically confirmed TB cases. Additionally, qXR reported an area under the curve (AUC) of 0.91–0.94 when benchmarked against radiologist interpretation [31, 32]. Integration of CAD with mobile digital X-ray vans increased case-detection yield by approximately 15–20% and reduced mean time-to-diagnosis by nearly 30% compared with conventional workflows [33]. Despite the prospects of AI technology for TB diagnosis, most evidence remains at the pilot scale and concentrated in urban or research-supported facilities [34]. Challenges include the authenticity and accountability of algorithm outputs, limited transparency of proprietary training datasets, and potential bias arising from under-representation of local radiographic patterns [35]. Implementation costs, including software licensing, device maintenance, and data-storage costs, are high, thereby limiting scalability [36]. Sustained funding, regulation, and cost-effectiveness analysis are integral to the sustainable deployment of AI-assisted radiographic screening in LMICs [36].
3.5.2. Portable Diagnostic Technologies
Mobile digital X-ray units integrated with artificial-intelligence software can increase access to TB screening in underserved and remote settings. Pilot projects in India, Indonesia, and Nigeria demonstrated that mobile X-ray vans equipped with CAD4TB or qXR software reduced the average time from initial screening to diagnosis from approximately 7 days to 2–3 days by enabling same-day interpretation and triage [37, 38]. These mobile units were particularly effective in reaching high-risk populations, such as urban slum residents, migrants, and mining communities, who hitherto lacked access to diagnostic services. In Nigeria and Indonesia, solar-powered X-ray systems with onboard batteries extended operational capacity in areas with unreliable electricity [39]. However, persistent connectivity issues limited real-time data transmission to national TB databases, and maintenance costs remained a significant barrier to scalability [40]. Evidence from pilot evaluations underscores that portable AI-enabled diagnostics can substantially shorten diagnostic delays and expand screening coverage when supported by robust infrastructure, digital integration, and local workforce training [41].
| Tool / Platform | Core Function | Countries Piloted / Implemented | Key Findings or Implementation Notes | Sources |
|---|---|---|---|---|
| CAD4TB (Computer-Aided Detection for TB) | Deep-learning algorithm for interpreting digital chest X-rays and triaging TB suspects | India, South Africa | Sensitivity 94–96%, specificity 77–83% vs. radiologists in pilot evaluations; improved community screening yield by 15–20% when combined with mobile vans | [24, 26, 27, 29] |
| qXR (Qure.ai) | AI-driven chest radiography interpretation using convolutional neural networks | India, Philippines | Accuracy comparable to expert readers; shortened average diagnostic time by ~30% in rural pilots | [25, 26] |
| 99DOTS | Phone-based medication adherence verification through missed-call/SMS logging | India, Uganda, Philippines | Low-cost solution; >85% adherence verification in early pilots; dependent on cellular coverage | [32-34] |
| Video-Observed Therapy (VOT) | Remote video check-ins to confirm dose ingestion | Indonesia, Pakistan | Comparable adherence outcomes to directly observed therapy (DOT); requires smartphone access and stable connectivity. | [10, 11] |
| Nikshay (India) | National digital case-based TB surveillance and adherence monitoring system | India | Integrates TB/HIV reporting and private-sector notifications; supports real-time dashboard analytics | [2] |
| AI Predictive Analytics Models | Machine-learning prediction of treatment default and adverse outcomes | Pakistan, Indonesia | Early pilots using demographic + clinical data achieved 80–85% predictive accuracy; support targeted CHW follow-up | [10, 37] |
3.5.3. AI Predictive Analytics and Treatment Monitoring
Artificial intelligence applications for tuberculosis management are being evaluated in pilot studies to support predictive analytics and treatment adherence monitoring. Early pilot programs in Pakistan and Indonesia developed machine-learning models using demographic, clinical, and adherence data to predict treatment default and complications [42, 43]. These models achieved predictive accuracies ranging from 80% to 85%, and enhanced community health worker (CHW) follow-up and the referral of patients at high risk of relapse and non-adherence. Mobile-based monitoring platforms such as 99DOTS and Video-Observed Therapy (VOT) utilized automated alerts and data dashboards powered by AI algorithms to flag missed doses and streamline supervision [44]. However, most initiatives remain in early development phases, with limited external validation and inconsistent data connectivity between facility-based and mobile systems [45]. Key barriers include the lack of interoperability among national TB databases, unreliable mobile network coverage, and limited local funding for digital technology innovations. Despite these limitations, AI-driven predictive analytics show potential to improve continuity of care, reduce loss to follow-up, and strengthen treatment outcomes when supported by reliable infrastructure, data governance, and programmatic integration [46]. A comparative summary of AI and digital-health tools currently implemented in high-burden LMICs is presented in Table 1.
Table 1 summarizes the central artificial intelligence (AI) and digital health tools currently being evaluated in low- and middle-income countries (LMICs). CAD4TB and qXR-enhance radiographic screening accuracy, while platforms such as 99DOTS, VOT, and Nikshay strengthen adherence monitoring and surveillance. Scalability is adversely affected by infrastructure constraints, training gaps, regulatory constraints, and funding gaps.
4. DISCUSSION
The findings of this review address each of the research questions posed in the introduction. First, persistent challenges in financing, human resources, and diagnostic coverage continue to limit the effectiveness of national TB programs. Second, AI-assisted diagnostics and digital adherence technologies have demonstrated strong potential for case detection and monitoring, but scalability is hampered by infrastructure and cost barriers. Third, policy and implementation analyses indicate that integrating TB programs into national health systems, investing in equitable workforce development, and establishing robust data-governance frameworks are integral to sustainable AI adoption in TB control. Collectively, these insights underscore the potential of AI to enhance TB programs when embedded within strengthened, equity-focused health systems. This integrative review highlights the need to reframe Tuberculosis (TB) control strategies in low- and middle-income countries (LMICs), where over 85% of global TB deaths occur annually [1]. India, Indonesia, Nigeria, the Philippines, and Pakistan remain at the epicenter of the epidemic, accounting for more than half of the global TB burden in 2022 [1, 2]. Despite decades of TB control efforts, LMICs continue to experience significant disparities in TB incidence, mortality, drug-resistant TB, and treatment outcomes. Structural weaknesses in health infrastructure, domestic funding, and social disparities are significant barriers to TB prevention, diagnosis, and treatment.
Following the reported high absolute burden of TB and multidrug-resistant TB (MDR-TB), India has made significant strides in implementing digital tools, such as the Nikshay e-platform, and in domestic resource allocation, covering over 80% of its TB program budget [2]. However, challenges persist with private-sector underreporting, regional health inequities, and delays in MDR-TB diagnosis and treatment [3]. Indonesia and Pakistan have demonstrated improved case detection and treatment success rates. However, they continue to rely significantly on international donors for TB financing, which may jeopardize sustainability and country ownership [4, 5]. TB mortalities in Nigeria are relatively high (76 per 100,000), with alarmingly high TB/HIV co-infection prevalence (21%). Nigeria's situation highlights the critical gaps in service integration and human resource deployment in high-prevalence, low-capacity settings [6]. Systemic and social determinants primarily contribute to the failure to achieve optimal TB control across the five LMICs. Gender disparities, socioeconomic vulnerability, urban slum conditions, and geographic inaccessibility. These contribute to delayed diagnosis and poor adherence [7, 8]. Researchers have found that Patients in rural Nigeria and parts of the Philippines travel multi-day journeys to health facilities to test and confirm diagnoses. Stigma and misinformation about TB and HIV have militated against early diagnosis and treatment [6, 9]. Delays in TB diagnosis among women are associated with caregiving burdens, financial disparities, and reduced autonomy in patriarchal societies in Nigeria and the Philippines [24, 45, 46]. These delays correlate with higher transmission rates and adverse clinical outcomes [23, 41]. Consequently, TB care must be conceptualized as a medical intervention and a holistic public health and development agenda.
AI tools, such as CAD4TB and qXR, validated in India and South Africa, demonstrated accuracy comparable to that of human readers in triaging chest radiographs [7,8,36]. However, few large-scale implementations exist outside pilot settings. Pakistan and Indonesia are exploring integrating AI-based predictive analytics to improve treatment adherence and predict default [10, 11, 37]. However, these tools require robust digital infrastructure, reliable internet connectivity and power supply, ethical and regulatory guidelines, and clinician acceptance to be effectively mainstreamed [12, 13]. With low digital readiness and limited internet penetration in rural areas, Nigeria has yet to meaningfully integrate AI into TB programs [6]. Nigeria’s low smartphone penetration, limited internet access in rural areas, and unreliable electricity supply continue to constrain the feasibility of Video-Observed Therapy (VOT) and mobile health applications [6, 38, 39]. Addressing these constraints is essential for digital equity in TB care.
This review underscores a pressing need for evidence-based, contextually grounded policy action. Domestic TB funding must be significantly increased, particularly in Nigeria, Pakistan, and the Philippines, where less than half of national TB budgets are self-funded [1]. Without substantial and reliable local funding, LMICs will continue to face funding gaps, program stagnation and discontinuation, and donor conditionalities. Strengthening domestic fiscal commitments, primarily through national health insurance schemes or performance-based allocations, would enhance accountability and sustainability [14, 15].
LMICs should prioritize the integration of TB services into primary health care, HIV/AIDS programs, maternal-child health services, and non-communicable disease platforms. This is particularly urgent in Nigeria, where fragmented vertical programs continue to undermine early case detection and co-management of TB and HIV [6,16]. Integrated service delivery can reduce stigma, improve continuity of care, and improve early diagnoses [17]. Investment in training, retaining, and equitably distributing health workers, particularly laboratory technicians, TB nurses, and community health officers, is paramount for implementing decentralized, patient-centered care models [18].
LMICs must scale up Digital and AI Technology through deliberate investment and cautious implementation of ethical, regulated deployment programs. National TB programs should adopt context-appropriate AI tools, based on operational research findings. For instance, deploying AI-enhanced CXR in mobile vans has improved case detection by up to 20% in rural communities in India [10]. However, intersectoral collaboration, sustainable local funding, health provider-led data protection, and participatory technology are needed to sustain these gains [12, 19]. The emerging literature on ethical AI, the cost-effectiveness of decentralized TB care, and gender-sensitive approaches further supports the need for multisectoral TB strategies tailored to local contexts [35, 38, 43, 46].
CONCLUSION
TB control in high-burden LMICs is beset with funding gaps, fragmented service integration (especially TB/HIV), and diagnostic capacity gaps. Evidence from pilot implementations demonstrates that AI-assisted radiography applications can augment and promote early detection, identify patients at risk of relapse, and monitor treatment adherence and outcomes when paired with infrastructure, training, and governance. Priority actions include increasing reliable and substantial domestic budget allocation, integrating TB across primary-care/HIV platforms, and implementing phased rollouts of AI tools and technologies with strong data-protection, ethical, and equity safeguards.
RECOMMENDATIONS
Domestic Financing: Increase national budget coverage (especially in Nigeria, Pakistan, and the Philippines) and embed TB within primary care and insurance benefits.
Service Integration: Co-locate TB with HIV, MCH, and NCD services; track cross-program metrics.
Diagnostics & Workforce: Expand GeneXpert/POC access; invest in TB nurses, lab techs, CHWs; strengthen digital literacy.
AI Scale-up: Phased adoption (mobile CXR + CAD; adherence tech), evaluate cost-effectiveness and real-world performance; implement ethical/data-protection frameworks.
Equity & Gender: Fund transport/cash-transfer supports; address gender-specific barriers and stigma.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contributions to the paper as follows: D.B.: Contributed to the study conception and design; B.A. and D.B.: Carried out data collection; B.A.: Responsible for the analysis and interpretation of the results; and B.A.: and D.B.: prepared the draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| AI | = Artificial Intelligence |
| CAD | = Computer-Aided Detection |
| CHW | = Community Health Worker |
| DAT | = Digital Adherence Technology |
| DOT | = Directly Observed Therapy |
| EBP | = Evidence-Based Practice |
| HIV | = Human Immunodeficiency Virus |
| LMICs | = Low- and Middle-Income Countries |
| MDR-TB | = Multidrug-Resistant Tuberculosis |
| NTP | = National Tuberculosis Program |
| POC | = Point of Care |
| PRISMA | = Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| RR-TB | = Rifampicin-Resistant Tuberculosis |
| TB | = Tuberculosis |
| VOT | = Video-Observed Therapy |
| WHO | = World Health Organization |
AVAILABILITY OF DATA AND MATERIALS
This study is a systematic review and did not generate any new datasets. All data analyzed in this review are from publicly available published sources, as cited in the manuscript. The authors did not collect primary or individual-level data.
ACKNOWLEDGEMENTS
Declared none.


