All published articles of this journal are available on ScienceDirect.
The Role of Herbal Supplements in Type 2 Diabetes Management: An Evidence from a Systematic Review
Abstract
Introduction/ Objective
Despite the increasing global prevalence of diabetes, herbal supplements are becoming increasingly popular as complementary therapies for the management of type 2 diabetes and its complications. This review examines their potential role in glycaemic control and diabetes care.
Methods
A thorough search was conducted across multiple databases, including the Cochrane Library, Scopus, PubMed, and Web of Science, supplemented by manual reviews to identify relevant studies. Key outcomes assessed were glycaemic control, lipid levels, safety, and metabolic markers. Study selection followed the PRISMA flow diagram, and data extraction was performed using a standardized, pilot-tested form. The Cochrane Risk of Bias Tool (RoB 2) was employed to evaluate study quality.
Results
Several herbs—such as cinnamon, fenugreek, ginger, and saffron—showed potential in improving HbA1c, fasting blood glucose (FBG), insulin sensitivity, and cholesterol levels, though some studies reported limited effects on glycaemic control. Curcumin, especially in nano-formulations or paired with piperine, significantly reduced FBG and HbA1c while enhancing β-cell function (HOMA-β). Fenugreek seed powder modestly lowered FBG and HbA1c, with more pronounced benefits on lipids. Ginger supplementation improved FBG, HbA1c, LDL cholesterol, and antioxidant status. Moringa yielded mixed results, with some studies noting postprandial glucose reductions but no significant glycaemic improvements. Saffron improved FBG and lipid profiles without notable effects on inflammation or oxidative stress.
Conclusion
They may serve as complementary treatments alongside conventional antidiabetic drugs, particularly for patients preferring natural options or those with mild to moderate T2DM. Future research should explore longer-term interventions and combination therapies for more definitive conclusions.
1. INTRODUCTION
Diabetes is a long-term health condition brought on by inadequate or inefficient insulin production [1]. Hyperglycaemia from uncontrolled diabetes can damage various body systems, particularly blood vessels and neurons [2]. In 1990, 7% of individuals aged 18 and over had diabetes; by 2022, that number had increased to 14% [3, 4]. The prevalence of untreated diabetes is highest in low- and middle-income nations, with more than half of persons with diabetes aged 30 and older not taking medication for their condition [5]. In 2021, diabetes was the cause of 1.6 million fatalities, with 47% of all deaths taking place in those under 70 [6]. It was also responsible for 11% of cardiovascular fatalities and 530,000 deaths from renal illness [7]. A balanced diet, regular exercise, maintenance of a healthy weight, and quitting tobacco use can all help prevent or postpone the onset of type 2 diabetes (T2DM) [8].
Poor glycaemic control and the subsequent micro- and macrovascular consequences of T2DM are the result of noncompliance with standard treatment regimens [9, 10]. Patients from ethnic minorities are more likely to have poor diabetes management, increasing their risk of morbidity, death, and disability from T2DM complications [11]. This might be partially explained by decreased health knowledge and lower adherence to conventional medications [12]. Despite having high blood glucose, some individuals may put off seeing a doctor or avoid using conventional medications because they are concerned about negative effects [13]. For instance, between 30 and 50 percent of patients are deterred from starting insulin because of fear, anxiety, or discomfort with injections [14].
Individuals of some ethnic minorities are more inclined to choose herbal remedies over conventional medical care [15]. In addition to standard medications, 50–60% of individuals with T2DM worldwide utilise some form of alternative therapy [16, 17]. Approximately two-thirds of South Asian participants in a UK study reported using herbal medications for a variety of conditions, including diabetes [18]. According to a previous systematic review, glycaemic management may be improved by the use of some herbal therapies in conjunction with conventional medical care [19]. However, most prior reviews have focused on single herbs or biochemical mechanisms in isolation, with limited integration of patient-centered or practice-oriented perspectives. The novelty of the present study lies in providing a comprehensive and qualitative synthesis of the most commonly used herbal remedies for T2DM, emphasizing their comparative effectiveness, dosage patterns, and contextual use across populations. The ultimate objective is to generate evidence-based, clinically relevant insights that can guide both patients and healthcare professionals in making informed decisions regarding herbal use for glycaemic management.
2. MATERIAL AND METHODS
2.2. Research Question
The review aims to address the following research question: “What is the efficacy and safety of herbal interventions (cinnamon, curcumin, fenugreek, ginger, moringa, saffron) in improving glycaemic control, lipid profiles, and other metabolic parameters in patients with T2DM?”
2.3. Eligibility Criteria
The PICO questions were assessed in this systematic review:
Population: Adult patients (≥18 years) diagnosed with T2DM.
Intervention: Any herbal intervention (e.g., cinnamon, curcumin, fenugreek, ginger, moringa, saffron) delivered in any form (e.g., capsules, powders, extracts).
Control: Placebo, routine care, or other active therapies.
Outcomes: Primary outcomes were glycaemic control (e.g., fasting blood glucose, glycated haemoglobin (HbA1c), insulin resistance) and lipid profiles (e.g., total cholesterol, LDL, HDL, triglycerides). Secondary outcomes included safety profiles (e.g., adverse events) and various metabolic markers (e.g., body weight, oxidative stress).
All randomised controlled trials (RCTs), including double-blind, triple-blind, and placebo-controlled studies, were included; studies published in English were included. While studies involving type 1 diabetes, gestational diabetes, or non-diabetic individuals. Non-randomised research, observational studies, case reports, and reviews. Studies with limited data or unclear methodology. Also, studies utilising combination therapy, where the impact of the herbal intervention could not be identified, and animal studies.
2.4. Search Strategy Databases
The strategy of this research was undertaken in multiple search engines and databases (the Cochrane Library, Scopus, PubMed, and Web of Science). The search method included a combination of MeSH terms and phrases linked to T2DM, herbal therapies, and outcomes of interest. Examples of search keywords include: “type 2 diabetes mellitus”, “herbal medicine”, “cinnamon”, “curcumin”, “fenugreek”, “ginger”, “moringa”, “saffron”, “glycaemic control”, and “lipid profile”.
2.5. Study Selection Screening
Two independent reviewers examined the titles and abstracts of all identified papers to determine eligibility based on the inclusion and exclusion criteria.
Full-Text Review: The complete texts of potentially eligible papers were retrieved and read separately by two reviewers. Disagreements were addressed by discussion or contact with a third reviewer.
The PRISMA flow diagram was used to describe the study selection process, with the number of studies identified and excluded for any reasons at each stage.
2.6. Data Extraction
Data Extraction Form: A standardised data extraction form was designed and pilot tested. Data were extracted separately by two reviewers, and differences were handled by consensus.
2.6.1. Extracted Data
Study characteristics: Author, year, country, study methodology, sample size.
Participant characteristics: Age, gender, and baseline glucose levels.
Intervention details: Type of herb, dose, duration, method of administration.
Comparator: Placebo or active control.
Outcomes: Glycaemic control (fasting glucose, HbA1c), lipid profiles (e.g., LDL, HDL), safety (adverse events), and other metabolic parameters.
In addition, the completed PRISMA checklist is provided as a supplementary file (Supplementary File 1).
2.7. Risk of Bias Assessment Tool
The risk of bias was assessed using the Cochrane Risk of Bias Tool (RoB-2) for RCTs. The assessed domains were bias originating from the randomisation method, bias owing to variations from desired interventions, bias owing to missing outcome data, bias in measuring the outcome, and bias in the selection of the reported outcome. Judgment: Each domain was rated as “low risk,” “some concerns,” or “high risk” of bias. The total RoB for each study and in summary was presented.
3. RESULTS
We obtained a total of 2223 studies, of which 1863 remained after removing duplicates. After reviewing the titles and abstracts, 964 items were removed, leaving 899 publications. During the full-text review, 865 papers were excluded. Finally, 34 studies fulfilled the inclusion criteria (Fig. 1).
Focusing on the use of several herbs for controlling T2DM, Table 1 presents a summary of the included studies’ characteristics [20-52]. It involves 28 studies conducted across several countries, including China, Brazil, Iran, Portugal, Thailand, Ethiopia, India, Saudi Arabia, Pakistan, Cameroon, and Algeria.
| First Author, Year | Country | Study Design | Overall Sample Size |
|---|---|---|---|
| Anderson, 2016 [20] | China | Placebo-controlled double-blind trial | 137 |
| Lira Neto, 2021 [21] | Brazil | Randomized triple-blind placebo-controlled trial | 140 |
| Mirfeizi, 2016 [22] | Iran | Triple-blind randomized clinical trial | 102 |
| Rachid, 2022 [23] | Portugal | Randomized controlled trial | 36 |
| Talaei, 2017 [24] | Iran | Double-blind randomized placebo-controlled trial | 39 |
| Zare et al., 2018 [25] | Iran | Triple-blind placebo-controlled randomized clinical trial | 140 |
| Adab 2019 [26] | Iran | Double-blind RCT | 80 |
| Asadi 2019 [27] | Iran | Double-blind randomized, parallel, placebo-controlled clinical trial | 80 |
| Hodaei 2019 [28] | Iran | Randomized, double-blind, placebo-controlled trial | 44 |
| Yaikwawong 2024 [29] | Thailand | Randomized, double-blind, placebo-controlled trial | 229 |
| Neta 2021 [30] | Brazil | Randomized double-blind placebo-controlled clinical trial | 61 |
| Panahi Yunes 2018 [31] | Iran | Randomized double-blind placebo-controlled trial | 100 |
| Rahimi H.R. 2016 [32] | Iran | Double-blind randomized placebo-controlled trial | 70 |
| Genet Alem Geberemeskel, 2019 [33] | Ethiopia | Experimental study using quantitative methods | 114 |
| Amit D. Kandhare, 2018 [34] | India | 12-week, randomized, double-blind, placebo-controlled, multi-center | 119 |
| Rania A Najdi, 2019 [35] | Saudi Arabia | Open-label randomized controlled trial | 12 |
| Narsingh Verma, 2016 [36] | India | Multicenter, placebo-controlled, double-blind, add-on clinical study | 154 |
| Suchitra, 2015 [37] | India | Randomized controlled trial | 60 |
| Arzati et al., 2017 [38] | Iran | Double-blind placebo-controlled trial | 50 |
| Carvalho et al., 2020 [39] | Brazil | Randomized double-blind clinical trial | 103 |
| Ghoreishi et al., 2024 [40] | Iran | Double-blind placebo-controlled trial | 76 |
| Khandouzi et al., 2015 [41] | Iran | Double-blind placebo-controlled trial | 41 |
| Shidfar et al., 2015 [42] | Iran | Double-blind placebo-controlled trial | 45 |
| Ahmad 2018 [43] | Pakistan | Randomized crossover | 20 |
| Taweerutchana 2017 [44] | Thailand | Randomized placebo-controlled | 32 |
| Sissoko 2020 [45] | Cameroon | Pilot clinical trial | 70 |
| Hameed 2023 [46] | Pakistan | Prospective placebo-controlled | 24 |
| Leone 2018 [47] | Algeria | Randomized controlled trial | 27 |
| Moravej Aleali, 2019 [48] | Iran | Double-blind randomized clinical trial | 64 |
| Azimi, 2015 [49] | Iran | Randomized controlled, single-blind, placebo-controlled clinical trial | 204 |
| Ebrahimi, 2019 [50] | Iran | Triple-blinded randomized clinical trial | 54 |
| Milajerdi, 2018 [51] | Iran | Triple-blinded randomized clinical trial | 54 |
| Shahbazian, 2019 [52] | Iran | Double-blind randomized clinical trial | 64 |
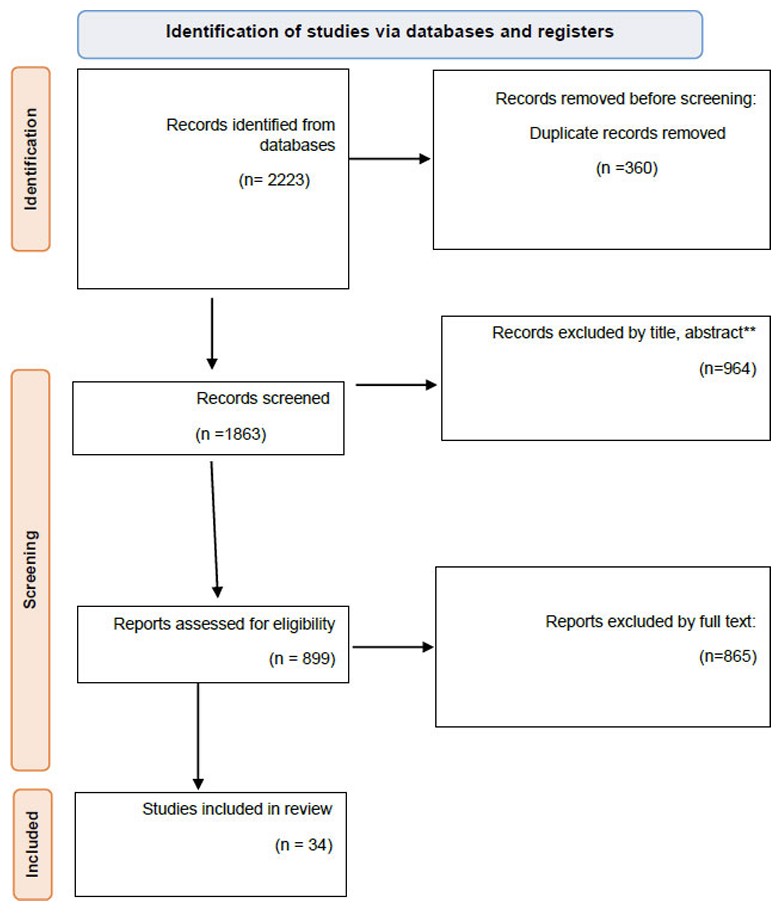
PRISMA flow diagram.
Out of 28 studies, 14 were carried out in Iran, highlighting the country’s important commitment to diabetes research and herbal remedies. Other countries with several included studies were Thailand (2 studies), India (3 studies), and Brazil (3 studies). Despite a focus on areas where traditional medicine is more common, this geographical variety points to a worldwide interest in herbal treatment for diabetes.
Considered the gold standard for clinical research, RCTs were the most common form of investigation. Most of these RCTs were double- or triple-blind, guaranteeing a significant degree of rigor in reducing bias. Some studies, such as Najdi et al. (2019), were open-label RCTs; although less precise than blinded studies, these methods can still offer insightful analysis [35]. Pilot clinical trials (Sissoko et al., 2020) and experimental investigations (Geberemeskel et al., 2019) evidence the use of a spectrum of methodological techniques, from exploratory to confirmatory research [33, 45].
Sample sizes varied widely throughout the research, ranging from as few as 12 (Najdi et al., 2019) to as high as 229 participants (Yaikwawong et al., 2024) [29, 35]. The majority of studies had sample sizes between 30 and 140 individuals, which is normal for clinical trials in this sector. However, smaller sample sizes (e.g., Hameed et al., 2023, with 24 participants) may restrict the generalisability of the findings; larger investigations (Yaikwawong et al., 2024) give more substantial evidence [29, 46].
Table 2 presents detailed information on the interventions and control groups of the included trials, concentrating on the exact herbal therapies, doses, durations, and control conditions employed in each experiment.
3.1. Herbal Interventions
The table highlights a range of studied herbal therapies, including cinnamon, curcumin, fenugreek, ginger, moringa, and saffron. Each plant was investigated in several trials, with variations in dosage and duration.
Cinnamon was the most frequently investigated herb, included in studies such as Anderson et al., 2016 (500 mg/day), Lira Neto et al., 2022 (3 g/day), and Zare et al., 2018 (500 mg capsules, twice a day) [20, 21, 25]. The dosages vary from 500 mg to 6 g per day, suggesting a wide range of studied quantities.
Curcumin (the active component of turmeric) was also frequently investigated, with doses ranging from 80 mg/day (e.g., Asadi et al., 2019) to 1500 mg/day (e.g., Hodaei et al., 2019, and Yaikwawong et al., 2024). Some research, such as Panahi et al. (2018), coupled curcumin with piperine (5 mg/day) to increase bioavailability [27-29, 31].
| First Author, Year | Intervention Group (Intervention & Sample Size) | Dosage | Duration | Control Group (Control & Sample Size) |
|---|---|---|---|---|
| Anderson, 2016 [ 20 ] | Cinnamon extract (64) | 500 mg/day | 2 months | Placebo (73) |
| Lira Neto, 2021 [ 21 ] | Cinnamon (71) | 3 g/day | 3 months | Placebo (69) |
| Mirfeizi, 2016 [ 22 ] | Cinnamon (27), Whortleberry (30) | 1 g/day | 3 months | Placebo (45) |
| Rachid, 2022 [ 23 ] | Cinnamon extract (18) | 6 g/100 mL | Single dose | Control (18) |
| Talaei, 2017 [ 24 ] | Cinnamon (20) | 3 g/day | 8 weeks | Placebo (19) |
| Zare et al., 2018 [ 25 ] | Cinnamon (70) | 500 mg capsules twice daily | 3 months | Placebo (70) |
| Adab 2019 [ 26 ] | Turmeric (40) | 2,100 mg/day | 8 weeks | Placebo (40) |
| Asadi 2019 [ 27 ] | Nano-curcumin (40) | 80 mg/day | 8 weeks | Placebo (40) |
| Hodaei 2019 [ 28 ] | Curcumin (21) | 1500 mg/day | 10 weeks | Placebo (23) |
| Yaikwawong 2024 [ 29 ] | Curcumin (114) | 1500 mg/day | 12 months | Placebo (115) |
| Neta 2021 [ 30 ] | Curcuma longa L. (33) | 500 mg/day with piperine 5 mg | 120 days | Placebo (28) |
| Panahi Yunes 2018 [ 31 ] | Curcuminoids (500 mg/day) + Piperine (5 mg/day) (n=50) | 500 mg/day curcuminoids + 5 mg/day piperine | 3 months | Placebo (n=50) |
| Rahimi H.R. 2016 [ 32 ] | Nano-curcumin (n=35) | 80 mg/day | 3 months | Placebo (n=35) |
| Genet Alem Geberemeskel, 2019 [ 33 ] | Fenugreek seed powder solution (57) | 25 g twice daily | 1 month | Metformin (57) |
| Amit D. Kandhare, 2018 [ 34 ] | IDM1 (60) | 700 mg three times daily | 12 weeks | Placebo (59) |
| Rania A Najdi, 2019 [ 35 ] | Fenugreek (6) | 2 g/day | 12 weeks | Glibenclamide (6) |
| Narsingh Verma, 2016 [ 36 ] | Fenfuro (77) | 500 mg bid | 90 days | Placebo (77) |
| Suchitra, 2015 [ 37 ] | Fenugreek (30) | Not clearly mentioned | Not mentioned | Control (Group C) |
| Arzati et al., 2017 [ 38 ] | Ginger (25) | 2000 mg/day | 10 weeks | Placebo (25) |
| Carvalho et al., 2020 [ 39 ] | Ginger (47) | 1200 mg/day | 90 days | Placebo (56) |
| Ghoreishi et al., 2024 [ 40 ] | Ginger (36) | 2000 mg/day | 3 months | Placebo (36) |
| Khandouzi et al., 2015 [ 41 ] | Ginger (22) | 2000 mg/day | 12 weeks | Placebo (19) |
| Shidfar et al., 2015 [ 42 ] | Ginger (22) | 3000 mg/day | 12 weeks | Placebo (23) |
| Ahmad 2017 [ 43 ] | Moringa (5% w/w) & SC (3% w/w) | Cookies | 1 meal | CC (20) |
| Taweerutchana 2017 [ 44 ] | Moringa leaf capsules (16) | 8 g/day | 4 weeks | Placebo (16) |
| Sissoko 2020 [ 45 ] | Moringa leaf powder (35) | 1g & 2g | 1 day | Control (35) |
| Hameed 2023 [ 46 ] | Moringa leaf capsules (12) | 3g & 6g | 3 months | Placebo (12) |
| Leone 2018 [ 47 ] | Moringa leaf powder (20 g) | 20 g | 1 meal | Control meal (27) |
| Moravej Aleali, 2019 [ 48 ] | Saffron (32) | 15 mg/day | 3 months | Placebo (32) |
| Azimi, 2015 [ 49 ] | Saffron (42) | 1 g/day | 8 weeks | Control (39) |
| Ebrahimi, 2019 [ 50 ] | Saffron (27) | 15 mg/capsule twice daily | 8 weeks | Placebo (27) |
| Milajerdi, 2018 [ 51 ] | Saffron (26) | 15 mg/capsule twice daily | 8 weeks | Placebo (26) |
| Shahbazian, 2019 [ 52 ] | Saffron (32) | 15 mg/capsule twice daily | 3 months | Placebo (32) |
Fenugreek was explored in studies like Geberemeskel et al. (2019) (25 g twice a day) and Najdi et al. (2019) (2 g/day), with varied doses and durations [33, 35]. Ginger was examined in dosages ranging from 1200 mg/day (e.g., Carvalho et al., 2020) to 3000 mg/day (e.g., Shidfar et al., 2015), with most research utilising around 2000 mg/day [39, 42]. Moringa was investigated in several forms, including capsules (e.g., Taweerutchana et al., 2017) and leaf powder (e.g., Sissoko et al., 2020), with doses ranging from 1 g to 8 g/day [44, 45]. Saffron was evaluated in dosages of 15 mg/day (e.g., Moravej Aleali et al., 2019) to 1 g/day (e.g., Azimi et al., 2015), with most research utilising lower quantities [48, 49].
3.2. Dosages and Durations
The doses of herbal therapies used varied widely between different studies, reflecting the variation in traditional and experimental applications of these plants. For example, cinnamon dosages ranged from 500 mg/day to 6 g/day, with doses between 500 mg and 3 g/day the most common. Curcumin doses ranged from 80 mg/day to 1500 mg/day, with some studies mixing it with piperine to increase absorption. Fenugreek dosages ranged from 2 g/day to 25 g twice daily.
There was also a large variation between the duration of treatments, from single doses (e.g., Rachid et al., 2022) to 12 months (e.g., Yaikwawong et al., 2024) [23, 29]. Most interventions lasted between 8 weeks and 3 months, suitable for testing short- to medium-term effects on glycaemic management and other metabolic parameters.
3.3. Control Groups
The majority of research used placebo-controlled designs, which are critical for assessing the efficacy of the herbal therapies. For instance, placebo groups were included in the designs of Anderson et al. (2016) and Lira Neto et al. (2022) to assess the effects of cinnamon extract and Adab et al. (2019) and Asadi et al. (2019) to examine the effects of curcumin and nano-curcumin, respectively [20,21.26.27]. Some research, such as Geberemeskel et al. (2019), instead used active controls (e.g., metformin) to test the efficacy of herbal remedies against conventional diabetic drugs [33]. A small number of studies, such as Rachid et al. (2022), employed non-placebo controls (control groups without any intervention), which may restrict the capacity to make significant conclusions about the efficacy of the herbal intervention [23].
3.4. Sample Sizes in Intervention and Control Groups
The overall sample sizes (Table 1) and those for control groups (Table 2) are presented to allow for an assessment of the balance between groups. For instance, Anderson et al. (2016) reported on 64 participants in the cinnamon group and 73 in the placebo group [20]. Lira Neto et al. (2022) had 71 participants in the cinnamon group, and in the placebo group, 69 participants [21]. Most investigations have somewhat balanced group sizes, which is critical for assuring the validity of the statistical comparisons. The characteristics of the study participants, such as their age, gender distribution, baseline blood glucose levels, and final blood glucose levels following intervention, are reported in Table 3.
3.5. Age of Participants
Different studies included a range of ages, from younger (Ahmad et al. 2017; mean age: 24.1 years) to older individuals (Anderson et al. 2016; mean age: 61.3 years) [20, 43]. The majority of studies used middle-aged to older participants, in line with the demographic typically affected by T2DM.
3.6. Sex Distribution
Sex distribution also varied between studies; some used a more balanced sample (e.g., Anderson et al. (2016), with 65.7% male participants) while others studied a predominantly female population (e.g., Yaikwawong et al. (2024), with 85% female participants) [20, 29]. In Rahimi et al. (2016), the intervention group (nano-curcumin) had a balanced gender distribution of 51.5% females, 48.5% males [32].
3.7. Initial Blood Sugar Levels
Most studies provide baseline glucose readings from participants (Table 3), indicating the severity of diabetes in the research populations. The large range of baseline glucose levels is a reflection of disparities in the severity of diabetes between patient populations and differences in inclusion criteria between studies. Baseline glucose levels ranged from 8.85 mmol/L (Anderson et al., 2016) to 123.65 mg/dL (Yaikwawong et al., 2024) and 180 mg/dL (Mirfeizi et al., 2016) [20, 22, 29]. It is difficult to assess the severity of diabetes across all included studies, as some, such as Geberemeskel et al. (2019), describe aberrant baseline glucose levels without using precise values [33].
3.8. Final Blood Sugar Levels
Most studies also provide the end glucose levels following the intervention (Table 3), allowing evaluation of the effects of herbal treatments on lowering blood glucose levels.
For example, Anderson et al. (2016) reported a small decrease in final glucose levels of 8.19 mmol/L [20]. Yaikwawong et al. (2024) recorded an average final glucose level of 115.49 mg/dL, significantly lower than the baseline [29]. Rahimi et al. (2016) reported a final mean glucose level of 118.38 mg/dL, which was lower than that of the placebo group [32]. Certain studies, such as Zare et al. (2018), suggest a drop in glucose levels based on the data but do not provide the final values [25].
3.9. Additional Baseline Features
Additional baseline variables, such as body mass index (BMI), lipid profiles, and insulin levels, are provided by certain studies and are crucial for comprehending the overall metabolic health of study participants. For instance, baseline insulin, cholesterol, and glucose values are reported by Panahi et al. (2018), while Rahimi et al. (2016) offer glucose levels with comprehensive baseline lipid profiles (TC, TG, LDL-C, and HDL-C) [31, 32].
| First Author, Year | Participants Age | Participants Gender | Baseline Glucose | Final Glucose |
|---|---|---|---|---|
| Anderson, 2016 [ 20 ] | 61.3 ± 0.8 years | 65.7% Male | 8.85 ± 0.36 mmol/L | 8.19 ± 0.29 mmol/L |
| Lira Neto, 2021 [ 21 ] | 62 years | 71.8% Female | 8.85 ± 0.36 mmol/L | 8.19 ± 0.29 mmol/L |
| Mirfeizi, 2016 [ 22 ] | 52-55 years | 11.1-30% Male | 180 ± 56 mg/dL | 155 ± 40 mg/dL |
| Rachid, 2022 [ 23 ] | 63.5 ± 1.6 years | 16.7% Male | 8.10 ± 0.75 mmol/L | 9.77 ± 4.58 mmol/L |
| Talaei, 2017 [ 24 ] | 57.6 ± 8.7 years | 38.5% Male | 183.85 ± 36.16 mg/dL | 172.20 ± 44.86 mg/dL |
| Zare et al., 2018 [ 25 ] | 30-80 years | Male and Female | FPG: 162.6±30.0 mg/dl | Not explicitly stated in the abstract, but implied to decrease |
| Adab 2019 [ 26 ] | 30-70 years | 51% Female, 49% Male | 133.79 ± 25.60 | 131.64 ± 28.33 |
| Asadi 2019 [ 27 ] | 30-60 years | Majority Female (87.5% female) | 165.7 ± 52.3 | 150.9 ± 58.1 |
| Hodaei 2019 [ 28 ] | 40-70 years | Majority Female (66.7% female) | 160 ± 35 | 153 ± 33 |
| Yaikwawong 2024 [ 29 ] | ≥35 years | Majority Female (85% female) | 123.65 ± 1.73 | 115.49 ± 1.70 |
| Neta 2021 [ 30 ] | 62.5 ± 11.0 years | Majority Female (77% female) | 203.9 | 197.3 |
| Panahi Yunes 2018 [ 31 ] | 18–65 years | 25 males, 25 females in both groups | 163 ± 37 mg/dL (curcuminoids), 174 ± 33 mg/dL (placebo) | 154 ± 34 mg/dL (curcuminoids), 171 ± 26 mg/dL (placebo) |
| Rahimi H.R. 2016 [ 32 ] | 56.34 ± 11.17 years (Nano-curcumin), 60.95 ± 10.77 years (Placebo) | Male: 17(48.5%), Female: 18(51.5%) (Nano-curcumin); Male: 14(40%), Female: 21(60%) (Placebo) | 135.5 ± 51.33 mg/dL (Nano-curcumin), 171.2 ± 50.0 mg/dL (Placebo) | 118.38 ± 11.95 mg/dL (Nano-curcumin), 146.32 ± 46.06 mg/dL (Placebo) |
| Genet Alem Geberemeskel, 2019 [ 33 ] | Newly diagnosed | Both sexes | Abnormal | Reduced significantly |
| Amit D. Kandhare, 2018 [ 34 ] | Mean age: 51.48 years | Both sexes | Abnormal | Reduced significantly |
| Rania A Najdi, 2019 [ 35 ] | >18 years | Both sexes | ≥140 mg/dL | Reduced insignificantly |
| Narsingh Verma, 2016 [ 36 ] | 25-60 years | Both sexes | ≤180 mg/dL | Reduced significantly |
| Suchitra, 2015 [ 37 ] | Not mentioned | Not mentioned | Not mentioned | Not mentioned |
| Arzati et al., 2017 [ 38 ] | 30 to 60 years old | Mixed | 189.4 ± 88.24 | 162.84 ± 64.10 |
| Carvalho et al., 2020 [ 39 ] | 20 to 80 years old | Mixed | 203.60 ± 88.24 | 174.05 ± 64.10 |
| Ghoreishi et al., 2024 [ 40 ] | 20 to 65 years old | Mixed | 174.5 ± 70.7 | 166.5 ± 68.1 |
| Khandouzi et al., 2015 [ 41 ] | 20 to 60 years old | Mixed | 161.50 ± 58.01 | 142.09 ± 47.90 |
| Shidfar et al., 2015 [ 42 ] | 20 to 60 years old | Mixed | 161.5 ± 58 | 142.09 ± 47.9 |
| Ahmad 2017 [ 43 ] | 24.1 ± 1.33 | 10 males, 10 females | Not specified | Not specified |
| Taweerutchana 2017 [ 44 ] | 55 ± 11 | Mixed | 138 ± 36 mg/dl | Not specified |
| Sissoko 2020 [ 45 ] | Diabetic: 45.8 ± 10.6 Nondiabetic: 50.1 ± 11.6 | Mixed | Diabetic: 140 ± 9 mg/dl Nondiabetic: 86.2 ± 8.4 mg/dl | Diabetic: 135 ± 11 mg/dl Nondiabetic: 84 mg/dl |
| Hameed 2023 [ 46 ] | 57 ± 9.2 | Mixed | 140 ± 9 mg/dl | 129.28 mg/dl |
| Leone 2018 [ 47 ] | For healthy: 42 ± 11 Diabetic: 62 ± 9 | Mixed | Healthy: 86.2 ± 8.4 mg/dl Diabetic: 315 ± 15 mg/dl | For healthy: 106 ± 3 mg/dl Diabetic: 296 ± 17 mg/dl |
| Moravej Aleali, 2019 [ 48 ] | Mean: 52.4±13 | Female: 24 (75%), Male: 8 (25%) | 173.2±73.9 | 147.9±53.5 |
| Azimi, 2015 [ 49 ] | Mean: 54.33±0.5 | Not specified | Not specified | Not specified |
| Ebrahimi, 2019 [ 50 ] | Mean: 54.59±7.09 | Not specified | 128.84±31.86 | 146.54±41.86 |
| Milajerdi, 2018 [ 51 ] | Mean: 53.5±9.9 | Not specified | 164.36±40.88 | 166.96±41.86 |
| Shahbazian, 2019 [ 52 ] | Mean: 53.5±9.9 | Female: 24 (75%), Male: 8 (25%) | 173.2±73.9 | 147.9±53.5 |
The main results and conclusions of the included studies are summarised in Supplementary 1, which focuses on how different herbal therapies affect lipid profiles, glycaemic management, and other metabolic parameters in individuals with T2DM.
3.10. Glycaemic Management
According to several included studies, postprandial glucose (PPG), HbA1c, and fasting blood glucose (FBG) levels significantly improved after herbal therapies. Anderson et al. (2016) reported that cinnamon extract enhanced insulin sensitivity (HOMA-IR) and decreased FBG by 0.66 mmol/L [20]. Yaikwawong et al. (2024) observed that curcumin supplements dramatically decreased HbA1c (6.12% vs. 6.47% in the placebo group) and FBG (115.49 mg/dL vs. 130.71 mg/dL in the placebo group) [29]. Meanwhile, Rahimi et al. (2016) showed that nano-curcumin decreased HbA1c by 0.9% and FBG by 17.12 mg/dL when compared to placebo [32]. However, other investigations, such as Talaei et al. (2017), found no effect of cinnamon supplements on glycaemic control (FBG, HbA1c, HOMA-IR) [24].
3.11. Lipid Profiles
Numerous studies document changes in lipid profiles, including increases in HDL-C and decreases in TG, TC, and LDL-C. According to Geberemeskel et al. (2019), fenugreek seed powder increased HDL-C (by 21.7%) while considerably lowering TC (by 13.6%), TG (by 23.53%), and LDL-C (by 23.4%) [33]. According to Neta et al. (2021), taking supplements of Curcuma longa L. increased HDL-C and decreased TG levels by 21% [30]. According to Shidfar et al. (2015), ginger supplements dramatically raised HDL-C levels and decreased LDL-C levels [42]. Nevertheless, certain studies, such as Kandhare et al. (2018), indicated that herbal therapies did not significantly alter lipid profiles [34].
3.12. The Function of Pancreatic β-cells and Insulin Sensitivity
Improvements in pancreatic β-cell function (HOMA-β) and HOMA-IR were reported in certain investigations. Yaikwawong et al. (2024) observed decreased HOMA-IR and enhanced HOMA-β after their curcumin intervention; however, this was contradicted by another study, which found no effect (Hodaei et al., 2019) [28, 29]. According to Panahi et al. (2018), curcuminoids and piperine decreased FBG levels and enhanced insulin sensitivity [31].
3.13. Oxidative Stress and Inflammation
Several studies showed decreases in oxidative stress and inflammatory markers after herbal therapy. According to Shidfar et al. (2015), ginger supplements raised antioxidant capacity (TAC) and decreased oxidative stress indicators (such as Malondialdehyde (MDA)) [42]. Moravej Aleali et al. (2019) reported that saffron enhanced antioxidant status and decreased inflammatory indicators (hs-CRP) [48]. However, Shahbazian et al. (2019) found no effect of saffron supplementation on inflammatory biomarkers like TNF-α or IL-6 [52].
3.14. Body Mass and Anthropometric Measurements
Following herbal therapies, some studies showed changes in waist circumference, body weight, and BMI. Adab et al (2019) found that turmeric supplements significantly lowered body weight and BMI [26]. Hodaei et al. (2019) observed decreased hip circumference and weight after curcumin treatment compared to placebo [28]. Nevertheless, other studies, including Talaei et al. (2017), indicated no discernible effects on body weight or BMI after cinnamon supplementation [24].
The conclusions varied widely between studies. Numerous authors concluded that lipid profiles, glycaemic management, and other metabolic parameters were improved by the specific herbal therapy given. Anderson et al. (2016) stated that in individuals with high blood glucose, cinnamon extract decreased FBG, insulin, and cholesterol levels [20]. Yaikwawong et al. (2024) reported that curcumin decreased FBG, decreased insulin resistance, and enhanced β-cell activity [29]. However, in other reports, such as Rachid et al. (2022), the herbal intervention had no discernible impact on glycaemic control (e.g., postprandial glucose response) or other measures [23].
3.15. Quality Assessment of the Included Studies
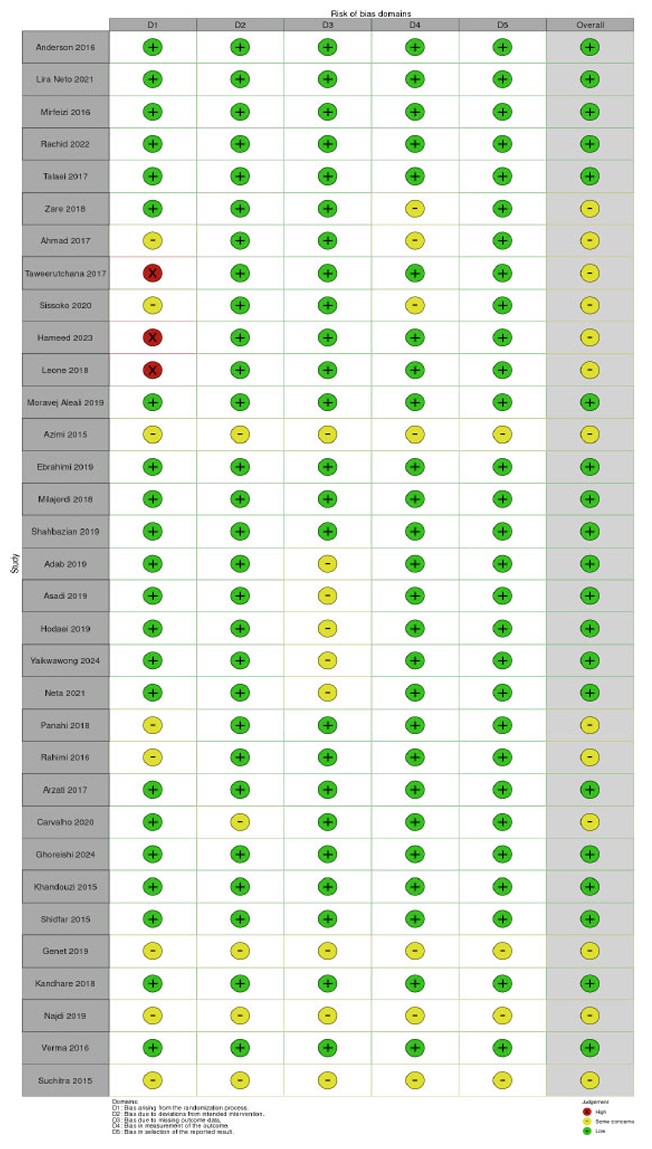
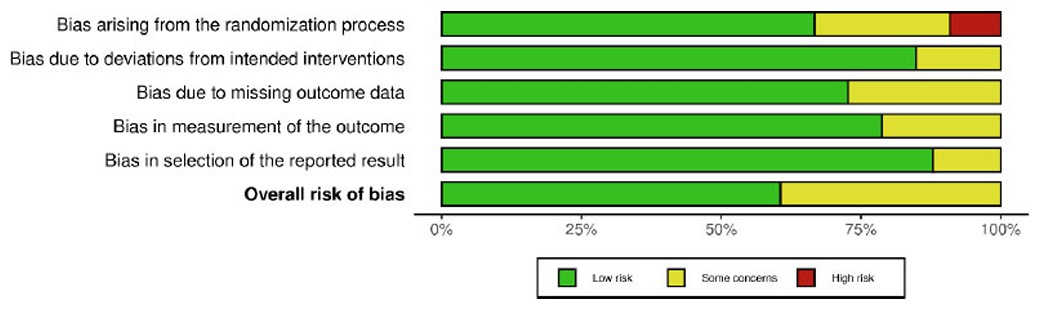
The risk of bias assessment identified five forms of bias: randomisation, variations from intended intervention, missing outcome data, assessment of outcome, and selection of reported results. Most studies exhibited minor risks of bias (Figs. 2 and 3). Green circles indicate a low risk in all domains. Some studies (Ahmad et al., 2017; Sissoko et al., 2020; Azimi et al., 2015; Rahimi et al., 2016; Verma et al., 2016; and Suchitra et al., 2015) exhibit moderate concerns, with yellow circles in one or more areas [32, 36, 37, 43-45, 49]. Domains D1 (randomisation procedure) and D4 (measuring of outcome) typically caused concern. A high risk of bias was detected for certain studies, with red markers suggesting a high risk in at least one category. These studies may have problems with randomisation or group allocation techniques (Taweerutchana et al., 2017; Hameed et al., 2023; Leone et al., 2018) [44, 46, 47]. Overall, most studies were deemed low risk, fulfilling essential methodological quality requirements. A few studies exhibited some concerns, and a few presented substantial risks in certain domains.
3.16. Quality Appraisal and Assessment
Upon the eleven items included for the quality of this study, the quality assessment showed that all studies addressed a clearly formulated research question, and their participants were randomised to the interventions. Suchitra (2015) did not report some of participant basic characteristics and baseline glucose levels. Besides, Azimi (2015), Ebrahimi (2019), and Milajerdi (2018) did not mention the gender distribution in the study, neglecting the gender effect on the study outcomes. Regarding the study duration, Yaikwawong's 2024 study continued the study for 12 months. with an including the probability of long-lasting effect of curcumin. While Rahimi H.R. 2016 took only 3 months of therapy, this may not give the long-lasting effect of the same drug. Because these studies tested a safe herbal product, the benefits of the experimental intervention outweigh the harms, neglecting the costs' effect. All studies with blind controlled trials affirmed the blindness, except for Rania A Najdi, 2019 [35], who reported an open-label study.
Risk of bias graph.
Risk of bias summary.
4. DISCUSSION
The study aimed to comprehensively investigate the efficacy of herbal therapies in controlling T2DM. Various included studies provided evidence that cinnamon, fenugreek, ginger, and saffron can improve FBG and HbA1c levels, increase insulin sensitivity, and lower cholesterol levels. However, others failed to show significant improvements in glycaemic management. Studies have suggested that cinnamon supplementation can significantly improve FBG and HbA1c levels, making it a valuable potential adjuvant for regulating blood glucose levels in individuals with T2DM [21, 53, 54]. Additional work has demonstrated its potential to boost insulin sensitivity and lower cholesterol levels, contributing to overall better metabolic health [53]. However, previous studies have also reported conflicting outcomes, finding no significant influence in these measures [24]. Factors contributing to variability include differences in study design, cinnamon dosage, intervention duration, and baseline characteristics of the study population.
Curcumin administration, particularly in nano-formulations or when mixed with piperine, has been suggested to provide significant improvements in FBG and HbA1c levels, demonstrating its potential as a valuable adjuvant in regulating blood glucose levels in T2DM patients. Other evidence suggests it can boost β-cell activity and lower insulin resistance, suggesting that it can strengthen the body’s ability to produce and respond to insulin. Additionally, curcumin has been proven to improve adiponectin levels, which are good for metabolic health, as they play a critical role in regulating glucose levels and breaking down fatty acids. Previous studies have also reported positive effects of curcumin on glycaemic control and metabolic health; nano-curcumin has been shown to significantly decrease HbA1c, FBG, and triglyceride levels in diabetic patients, and to prevent high-fat diet-induced insulin resistance and obesity by attenuating lipogenesis in the liver and inflammatory pathways in adipocytes [32, 55, 56]. Overall, curcumin shows potential as an adjuvant herb for glycaemic control and metabolic health.
Reductions in FBG and HbA1c levels with fenugreek seed powder intake indicate a favourable effect on glycaemic control [57]. Fenugreek seed powder also demonstrated significant changes in lipid profiles, lowering TC, LDL-C, and TG levels while elevating HDL-C; each of these is beneficial to cardiovascular health. Previous studies have indicated similar effects on lipid profiles of hypercholesterolemic T2DM patients [58]. Other studies have indicated more significant results, such as Shabil et al.'s (2023) systematic review, which found a drop in FBG and HbA1c levels with fenugreek intake, although results varied among trials [59]. Fenugreek seed powder shows potential for improving glycaemic management and lipid profiles, with more consistent results for cholesterol-lowering benefits. However, the diversity in glycaemic control outcomes underscores the need for additional standardised studies to find the ideal circumstances for its administration.
Ginger supplementation has been demonstrated to improve glycaemic management, lipid profiles, and antioxidant capacity, and reduce oxidative stress in persons with T2DM. Studies have demonstrated that ginger can dramatically lower FBG, HbA1c, and LDL cholesterol levels, suggesting it can be effective for regulating blood glucose levels and improving lipid profiles [38]. Additionally, ginger supplementation has been demonstrated to boost antioxidant capacity and lower oxidative stress markers, such as MDA. Previous studies have also indicated comparable advantages, showing significantly lowered TC, LDL-C, and TG levels [60]. However, studies have shown inconsistent results on oxidative stress, with some indicating considerable reduction in inflammation and oxidative stress indicators [61, 62].
Research on the impact of Moringa oleifera on glycaemic management has shown inconsistent results. Some studies have observed decreases in postprandial glucose levels, suggesting that Moringa may help regulate blood sugar levels after meals [63-65]. However, Taweerutchana et al. (2017) showed no significant effects on overall glycaemic measures, demonstrating variability in the efficiency of Moringa for blood glucose control [44]. Moringa oleifera has potential benefits for blood pressure control, as its leaf extract contains chemicals, including flavonoids and potassium, which have antihypertensive properties [66]. Studies have demonstrated that Moringa leaf extracts have considerable free radical scavenging activity and can increase the body’s antioxidant defences [67, 68].
Studies have demonstrated that saffron supplementation can enhance FBG and lipid profiles, potentially assisting T2DM patients in regulating blood glucose levels and improving lipid profiles. However, these studies also revealed no significant influence on inflammatory markers or oxidative stress, suggesting a limit to saffron’s benefits in this regard. Previous studies have revealed similar positive effects of saffron supplementation on glycaemic control and lipid profiles [69, 70]. However, other studies have produced inconsistent results on inflammatory markers and oxidative stress, with some indicating favourable effects while others have no significant influence [50]. Abedi et al. (2023) revealed that saffron supplementation significantly decreased MDA and total oxidant status (TOS) levels while raising total antioxidant capacity (TAC) and glutathione peroxidase (GPx) levels [71].
5. LIMITATIONS OF THE STUDY
The systematic review and meta-analysis on herbal interventions for T2DM have some limitations, including heterogeneity across studies and varying doses and durations of treatment. The short length of many of the therapeutic interventions also restricts the ability to examine long-term effectiveness and safety. Limited evidence on safety and adverse effects was provided in many studies, further hindering evaluation, particularly of long-term safety. Likewise, numerous studies included were executed with comparatively small sample sizes, thereby constraining the generalisability of their findings and augmenting the likelihood of statistical uncertainty. The brief period of interventions in certain studies may have been inadequate to comprehensively assess long-term effects or the sustainability of the observed outcomes. These issues must be taken into account while evaluating the overarching results of this review. Future research with larger, more powerful samples and longer follow-up periods is required to confirm and expand on these findings.
CONCLUSION
This systematic review concluded that cinnamon supplementation significantly reduced FBG, HbA1c, and insulin sensitivity. Curcumin, particularly in nano-formulations or coupled with piperine, demonstrated substantial improvements in FBG, HbA1c, and β-cell activity. Fenugreek was beneficial in lowering lipid profiles, whereas ginger supplementation significantly lowered FBG, HbA1c, and LDL-C. Moringa lowered postprandial glucose levels, but had no significant effect on overall glycaemic control. Saffron enhanced FBG and lipid profiles but had no significant effect on inflammatory markers or oxidative stress.
Herbal therapies can be considered as supplementary therapies for conventional diabetic medications, particularly for individuals seeking natural options or those with mild to severe T2DM. Further research should focus on extended intervention durations and combination therapies.
AUTHOR'S CONTRIBUTIONS
The sole author conducted the entire work, including conceptualization, literature search, data extraction, analysis, manuscript writing, and final approval.
LIST OF ABBREVIATIONS
| TC | = Total Cholesterol |
| TG | = Triglycerides |
| LDL-C | = Low-Density Lipoprotein Cholesterol |
| HDL-C | = High-Density Lipoprotein Cholesterol |
AVAILABILITY OF DATA AND MATERIALS
The data and supportive information is available within the article.
ACKNOWLEDGEMENTS
The author would like to acknowledge Nawaf Al Khashram and Mohammed Bu Bshait for their assistance in reviewing and organizing articles during the initial stages of this systematic review.


